By Molnár-Institute for…
Study shows simultaneous DryLab®4 modeling can overcome problems with robustness originated from mobile phase effects in HPLC
Berlin: – Three scientists from Molnár-Institute for Applied Chromatography have contributed to a newly-published research study that demonstrates how various liquid chromatography dependencies can be modelled simultaneously in silico using the institute’s DryLab®4 software platform for significant robustness and productivity gains.
The study ‘Simultaneous optimization of mobile phase composition and pH using retention modeling and experimental design’ published in the Journal of Pharmaceutical and Biomedical Analysis is lead authored by Dr. Norbert Rácz, former PhD student at the Department of Analytical Chemistry at Budapest University of Technology, assisted by Molnár-Institute founder and President, Dr. Imre Molnár and colleagues Dr. Arnold Zöldhegyi and Dr. Hans-Jürgen Rieger. The team also included Dr. Róbert Kormány from Budapest-based Egis Pharmaceuticals Plc.
Improving mobile-phase-related robustness
Against a background of increasing sample complexity and safety expectations leading to higher regulatory demands, the team set out to investigate the impact of mobile phase influences as important factors of selectivity tuning in method development in liquid chromatography. Their goal was to develop a method that would mitigate mobile-phase-related robustness issues throughout the method lifecycle.
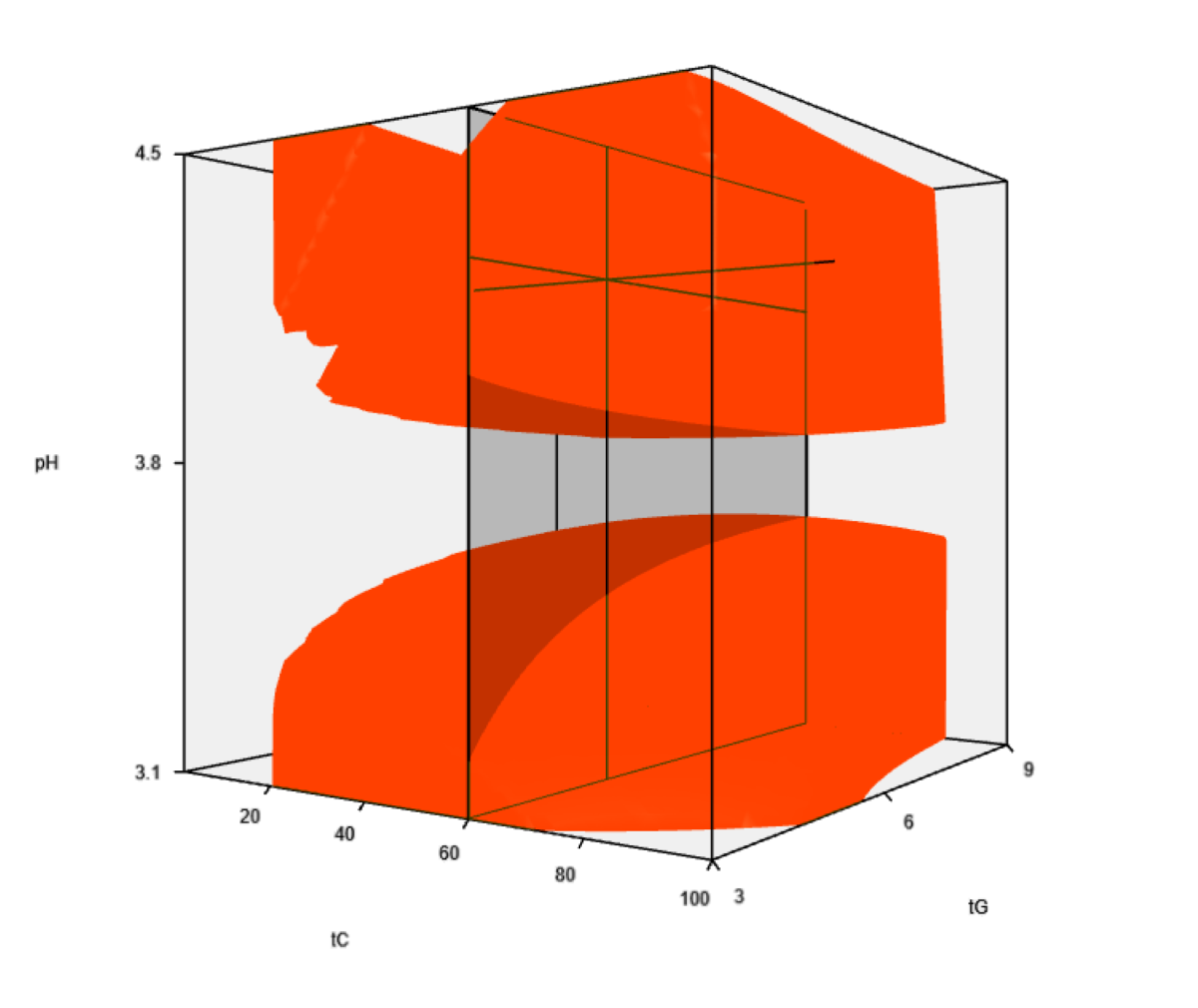
To discover and understand these effects, they tried a new module of chromatographic modeling software DryLab®4 (ver. 4.3.4. beta) along with a special experimental design (DoE) that would allow simultaneous optimization of eluent-dependent parameters, such as gradient time (tG), ternary eluent composition (tC) and pH, requiring 18 input experiments tG-pH-tc (2 × 3 × 3 = 18).
Excellent agreement with prediction
The model’s creation, based on a UPLC system of Waters and narrow bore column (50 × 2.1 mm), allowed the entire experimental work to be finished in under three hours. To demonstrate the applicability of this new design, amlodipine and its related pharmacopoeia impurities (A–H) were subjected to be used in a case study.
Predicted vs. Experimental (or Verification) runs showed excellent agreement, average retention time deviations were typically around one second. Modelled robustness testing was also performed, elucidating all important mobile phase and instrument parameters that could influence a method’s lifetime performance. Furthermore, as the in silico robustness testing is the least time consuming part of the method development process, the researchers found it could be used extensively to evaluate robustness even at the very early part in stage 1 of the Method Life Cycle (MLC).
Creating multifactorial design space
“The novelty of the work was providing a scientific way to visualize these dependencies in a highly variable multifactorial design space. In addition, compiling all possible combinations of solvent- (tC, pH), and instrument-dependent (tG, flow rate, start[B%], end[B%]) factors, specified with their tolerance limits, modeling allowed an integrated early-stage multi-parameter robustness characterization,” the study concludes.
“This, in the light of scarcity of relevant regulation guidelines, can effectively support the analyst to design methods with long-term separation quality, already at the development phase,” the authors add.
“For the last, this real life-related case study was a perfect example on how to implement an early stage robustness, with a particular interest in solvent effects on selectivity. This we believe, can contribute to a better control strategy of mobile phase, and from instrument dependent chromatographic parameters. This can safeguard methods, regarding expected performance across all stages of the lifecycle,” the study finds.
About MOLNÁR-INSTITUTE
Founded in 1981, Molnár-Institute develops DryLab®4, a software for UHPLC modeling for a world-wide market. Its powerful modules allow for the most sophisticated method development as required across pharma industries. Analytical scientists use DryLab® to understand chromatographic interactions, reduce runtimes, increase robustness, and conform to Analytical Quality by Design (AQbD) standards.
The Molnár-Institute is a registered vendor to the US FDA, CDC and many other regulatory bodies. DryLab® has pioneered AQbD long before regulatory agencies across the world encouraged such submissions. Widely implemented by thought leaders, the software contributes substantially to the paradigm shift towards a science and risk driven perspective on HPLC Quality Control and Assurance.
Further information at: http://www.molnár-institute.com/
Resources
Click on Simultaneous optimization of mobile phase composition and pH using retention modeling and experimental design to access full study.