By Molnár-Institute for…
MOLNÁR-INSTITUTE highlights ICH Q14 challenges to high performance liquid chromatography
Berlin: – The Molnár-Institute for Applied Chromatography has identified the recently published International Council for Harmonization ICH Q14 Quality Guidelines as a potential ‘game changer’ for the science of biochemical analysis.
The Q14 guidelines not only demand researchers to describe methods and show their robustness, but also recommend that they provide rationale for choosing that method in the first place and how it was developed in that particular way.
Emphasis on Design Space
Meeting Q14 guideline unbraces unmethodical development practice (trial and error) and emphasizes science-driven risk assessment and risk management. This goes beyond the scope of simple statistics and necessitates the establishment of meaningful design-of-experiments (DoE) options along with powerful mechanistic modelling engine to effectively support the creation of a fully modelled, comprehensive Design Space (DS).
Molnár-Institute founder and President, Dr. Imre Molnár, commented: “Some packages are statistically based and they will run plenty of experiments, keeping your instrumentation busy, day-in, day-out, in an automated way. A statistical package will deliver an incomplete DS-knowledge and therefore will only able to tell you, if your method works or if it fails, but it won’t demonstrate what exactly and WHY is something happening in your Design Space.”
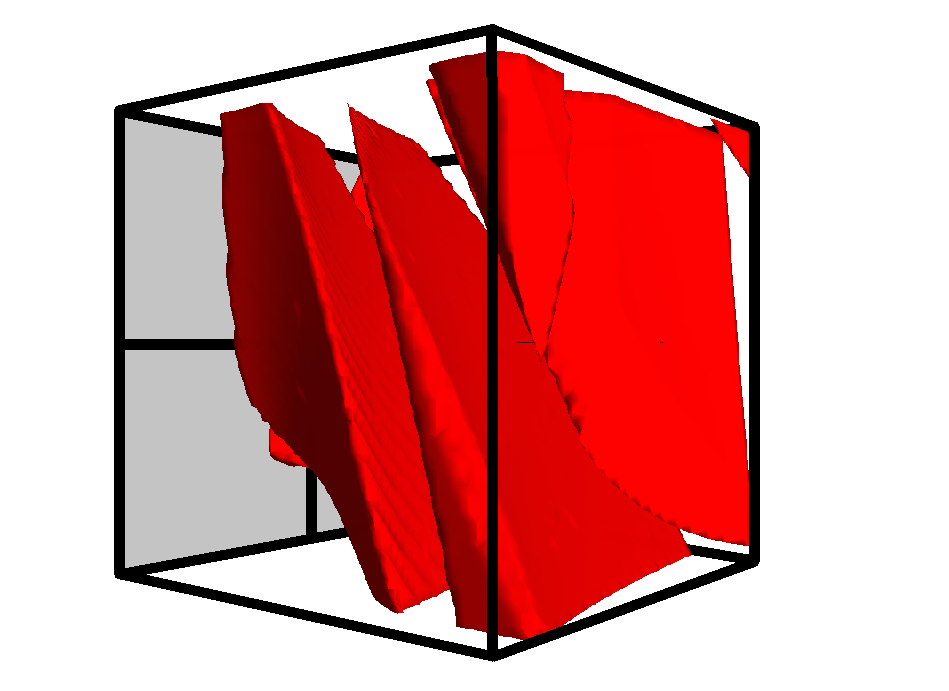
“DryLab® adopts a different, sound chromatographic science-based approach to visualize all the relevant interactions of any separation at all possible method conditions — so that researchers can precisely see, what areas their method will work in, which parameters can be varied, and which need to be strictly controlled,” he noted.
Connecting the dots
DS analysis is key to rationalizing HPLC and UHPLC-methods. Molnár-Institute’s DryLab®4 software modelling platform is one of the few in the world, that by its design concept, will fully comply with ICH Q14 requirements in being able to illuminate a total DS, that could typically comprise more than a million modelled workpoints or method-parameter combinations and has repeatedly proven potential to revolutionize pharmaceutical chemistry and research.
“The main benefit of DryLab® is, that it requires only a minimum number of experiments (two or three per chosen parameter) to accurately model the underlying phase system. Therefore, in the case of a 3-dimensional-DS, only 12 distinct experimental input runs are needed to discover and model all of the possible chromatographic interactions. It’s like connecting the dots, only you need to know, how to connect them, this is what DryLab® exactly does.” Dr. Molnár explained.
Flexible Design Choices
Dr. Molnár’s observations are supported by a recent research-based article ‘Flexible Design Choices’ authored by a team from the Institute headed by application specialist, Arnold Zöldhegyi of the Molnár-Institute-Team, and recently published by the online journal Chromatography Today.
The article explores the implications of Design Space Modelling in yielding faster production and cutting time-to-market, with comparison of DS-based models allowing for more science-driven decision-making and risk-assessment, potentially eliminating all out-of-specification (OoS) investigations. The authors argue, that collection of retrieved data enables creation of Knowledge Management Reports that can also incorporate method robustness evaluation, resulting in shortened submission processes and more transparent communications with regulatory agencies.
Regulatory implications
The article notes, that modern analytical methods must be highly adaptable to Lifecycle Management and must always deliver high performance, with the whole pharmaceutical industry transitioning towards Analytical Quality by Design (AQbD) approaches.
“An aspect of AQbD is to include tolerance limits of the parameters involved, along with other systematic elements, such as a Design of Experiments (DoE) creating each Design Space,” the article notes.
“In the past, Analytical Quality-by-Design principles were adopted from the manufacturing Quality-by-Design guides, thus the corresponding terminologies and detailed steps remained vague and were interpreted and implemented differently by independent groups. However, one can now turn on the terminologies and principles used in the draft ICH-Q14, which will ask, why a method was developed in a particular way and not otherwise. Another guide from the USP <1220> on Lifecycle Management is also pointing in that direction,” as the authors point out.
Method Lifecycle Management
USP <1220> aims to substitute the traditional way of method development and validation, with its high number of wet-lab experiments tied to studied data points, with a more structured and holistic approach introducing Method Lifecycle Management. This new approach integrates existing chromatographic knowledge as derived from fundamental theories.
“Such mechanistic modelling by design offers a thorough, holistic understanding and allows for unparalleled modelling versatility. The relevant chemometrical chapter of USP <1039> also highlights the use of mechanistic models,” the team observes.
Evolved DS Modelling
The article describes an evolved form of DS modelling, based on orthogonal peak tracking and using Mass Spectrometry, where available, to present additional peak information to the analyst in addition to the optical detection data — UV-trace most commonly. After the assessment of the model’s robustness, confirmation runs around the setpoint are seamlessly transferred to Waters Corporation proprietary chromatographic data system Empower, where they are executed to be retrieved by DryLab® afterwards.
“By implementing such automated workflows, the reliability and throughput can tremendously be increased, while human, inter-operator errors can be eliminated,” the article explains.
The team observed: “The described DryLab® Design Space Modelling approach moves away from traditional method development routines based only on statistics. Instead, and in line with the recent ICH Q14 draft and USP<1220>, the model yields a profound understanding of the chromatographic interdependencies within the separation system while a Holistic Control Strategy (HCS) can be derived from it.
“Design Space Modelling effectively expands the chromatographer’s skillset in his quantitative analytical work and can be implemented on a daily basis using the seamless connection of software packages, in this case DryLab® and Empower. By implementing automated workflows, the development of new and urgently needed pharmaceuticals can be developed with much faster time to market – to the advantage of the suffering patient,” the article concludes.
About MOLNÁR-INSTITUTE
Founded in 1981, Molnár-Institute develops DryLab®4, a software for UHPLC modelling for a world-wide market. Its powerful modules allow for the most sophisticated method development as required across pharma industries. Analytical scientists use DryLab®4 to understand chromatographic interactions, reduce analysis time, increase robustness, and conform to Analytical Quality by Design (AQbD) principles, according to ICH Q14.
The Molnár-Institute is a registered partner to the US-FDA, CDC and many other regulatory bodies. DryLab®4 has pioneered AQbD long before regulatory agencies across the world encouraged such submissions. Widely implemented by thought leaders, the software contributes substantially to the paradigm shift towards a science and risk driven perspective on HPLC Quality Control and Assurance.
Further information at: http://www.molnar-institute.com/
Resources
Click on Flexible Choices in Design Space Modelling to Support Smooth Pharma Production to access original article.
Click on ICH Q14 Guidelines for further information.