By Molnár-Institute for…
Molnár-Institute analytical method development and validation
Molnár-Institute has eased analytical method development and validation in high performance and ultra-high performance liquid chromatography (HPLC/UHPLC) by incorporating QbD based analytical method development into its proprietary software suite, DryLab®.
The current iteration, DryLab®4, provides a state-of-the-art tool to support analytical scientists in HPLC/UHPLC method development and validation, predicting chromatograms in silico under a much wider range of experimental conditions than would be possible in the laboratory and compiling data in forms that ease the task of preparing technical acceptance documents.
Analytical method selection essentials
Effective analytical method development is essential in pharma HPLC/UHPLC processes to predict and manage availability of the various columns, operational parameters, mobile phase composition and values for pH of eluents.
Analytical method selection follows a six-step basic process:
- Select an HPLC/UHPLC analytical method: defining the method for preparation of sample to produce a clear solution for chromatography, respecting particular solubility, filtration and extraction requirements.
- Chromatographic method: While most samples can use Reversed Phase Chromatography (RPC),
some will require ion suppression for weak acidic or basic samples on Reversed Phase with ion
pairing for strongly polar samples. Other stationary phases could also be applied (such as cyano
bonded-, ion exchange, or HILIC-phases, etc.) - Select gradient or isocratic HPLC: The third step is to make the choice between the greater resolution of gradient HPLC or the wider bandwidth of isocratic HPLC.
- Column size: Selection of column size with flow rates and column particle size reflects sample complexity with 50 – 100 mm columns used for most samples, but longer columns up to 150 mm needed for more complex samples.
- Detectors: Choice also need to be made between UV, fluorescence, electrochemical or refractive index HPLC detectors, depending on chromophore content, trace analysis needs or concentration levels.
- Wavelength: Finally, wavelength has to be optimised to provide the ideal balance between
sensitivity and noise elimination.
Analytical method development steps
Once the analytical method is fully defined, it will be necessary to select different chromatographic conditions, matching the flowrate to solvent concentration in the mobile phase, with concentration optimised to the desired retention time and capacity factor.
Finally, sample runs are used to tune parameters, including column and particle sizes, run times, flowrates to achieve optimal resolution and shortest run times.
Once the analysis method is optimized, it also needs analytical method validation for consistency, now a mandatory requirement within all regulatory agencies.
ICH guidelines for analytical method validation
International Council for Harmonization (ICH) guidelines for method validation define the nine principal pharmaceutical validation characteristics that must be addressed in technical specification documents:
- Analytical Procedure: detailed description of the steps necessary to perform each analytical test.
- Specificity: the ability to assess unequivocally the analyte in the presence of expected components, including degradants, matrix components, etc.
- Accuracy: also termed ‘trueness’, this expresses the closeness of agreement between the value, which is accepted either as a conventional true value or an accepted reference value and the value found.
- Precision: the closeness of agreement (degree of scatter) between a series of measurements obtained from multiple sampling of the same homogeneous sample, considered at three levels: repeatability, intermediate precision and reproducibility.
- Detection limit: the lowest amount of analyte in a sample that can be detected.
- Quantitation limit: the lowest amount of analyte in a sample that can be quantitatively determined with suitable precision and accuracy.
- Linearity: ability within a given range to obtain test results directly proportional to concentration of analyte in the sample.
- Range: the interval between the upper and lower concentrations of analyte in the sample for which it has been demonstrated that the analytical procedure has a suitable level of precision, accuracy and linearity.
- Robustness: measure of the procedure’s capacity to remain unaffected by small, but deliberate variations in method parameters, indicating its reliability during normal usage.
The DryLab software makes it easier to establish the correct ranges and to quantify all these method validation parameters to comply with analytical method validation guidelines.
DryLab analytical method selection
Researchers can use DryLab to determine quickly and easily exactly, how separations behave, as they simultaneously vary multiple method parameters, such as pH, temperature, buffer concentration, or ternary eluent composition, (tC), etc.
DryLab thus offers considerable speed, efficiency and cost advantages to any organisation developing HPLC methods that wish to optimize the separation of complex samples.
DryLab HPLC analytical method development
DryLab can use data generated from as few as two input experiments to predict resolution and retention times for millions of unique, virtual chromatograms.
The researcher can define a chosen analytical target profile (ATP) and then rely on DryLab to shape a systematic approach and establish initial method conditions.
Required input experiments can be uploaded in the universal format as *.cdf-files, available under “Export” in most chromatographic data systems (CDS) importing them into DryLab, helping to manage input runs, streamline peak tracking and exclude potential errors.
Following the data import, DryLab will generate a matched peak table, ready to build the HPLC model.
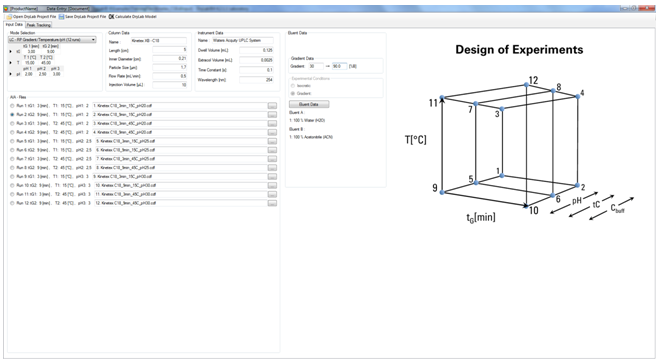
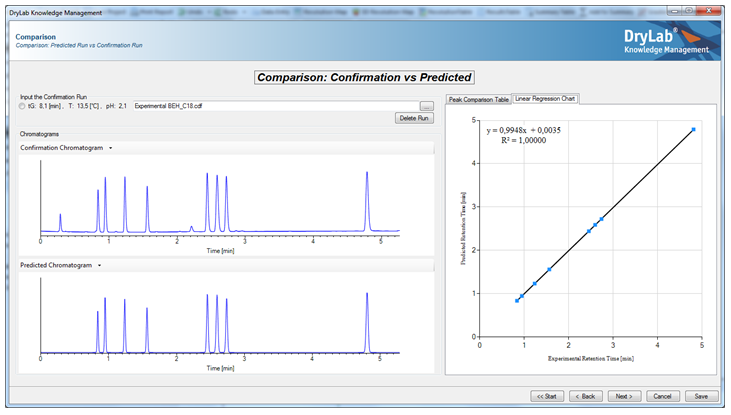
Based on initial Design of Experiment (DoE), DryLab will create multi-dimensional resolution models that depict the set of conditions to achieve baseline separation or higher. Predicted chromatograms are viewable in seconds for any point within the model. A single mouse click can select the working point with the highest critical resolution. DryLab can predict resolution and retention times with very high accuracy.
Advanced functions
DryLab allows to predict, how changes in additional method parameters will affect the separation, such as flow rate, column dimensions, instrument parameters, and eluent %B in gradient and in isocratic conditions. This provides maximum flexibility for method optimization and analytical method transfer.
Once initial working point is selected, DryLab can test the tolerance limits of a separation by evaluating the impact of small fluctuations on up to 4 measured and 8 calculated critical separation parameters. This allows researcher to determine which parameter exert the greatest influence on the separation quality and therefore must be strictly controlled to achieve complete success in routine applications.
Finally, the researcher can create a GMP compatible Method Documentation as a comprehensive Knowledge Management Document, that represents a method validation protocol by compiling a complete set of method data directly from DryLab to provide a platform from which the user and regulators are able to justify and comment on method criteria and choices. This report is GMP compliant and is the perfect tool for better knowledge sharing.
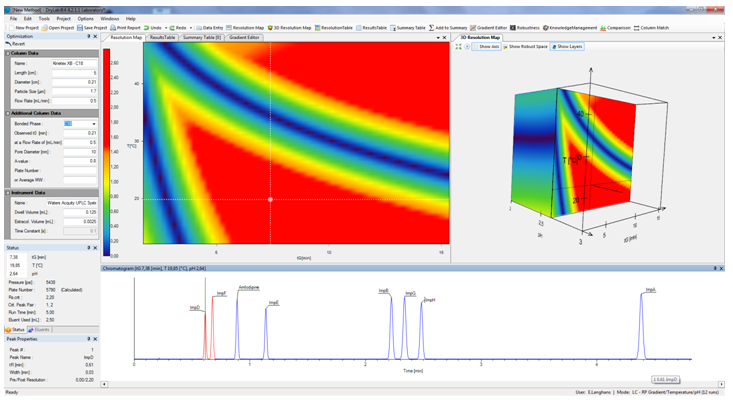
DryLab uses real data to create color-coded critical resolution maps, plotting critical resolution as a function of 1, 2 or 3 selected critical method parameters. In addition to visualizing the interaction of these parameters, it can also predict chromatograms for changes in other method conditions, such as column dimensions, flow rate, gradient elution, instrument parameters, etc.
Additional Features & Modules
In addition, DryLab includes a number of advanced functions to increase the value of HPLC models.
- Peak Tracking: Identification and assignment of peaks from a set of systematic experiments is an important first step in controlling the HPLC method development and achieving process validation.
- Gradient Editor: This powerful tool optimizes the separation via gradient elution, modifying gradient time, changing start and end %B, and adding further gradient steps. This process can either be controlled manually or rely on DryLab to find the best linear or step gradient automatically.
- ColumnMatch: This tool provides comparison between different columns, taking into account contributions such as hydrophobicity, steric selectivity, hydrogen-bond-acidity, hydrogen-bond-basicity, and ion-exchange properties of the silanol groups at different pH-values. Selecting columns that are very different in their selectivities can reveal hidden peaks.
- 3D-Module (the “Cube”): DryLab’s Cube extends the 2D resolution map into the third dimension, establishing the Method Operable Design Region (MODR) visualizing the influence of three parameters in which researcher can explore multifactorial variabilities for the most robust separation and for HPLC method validation. In addition, it can model up to eight other parameters, including column dimensions, flow rate, gradient points, and instrument parameters. The Cube generates exponentially more conditions in which to model the method and offers an intuitive display of how simultaneous changes to multiple method parameters affect the critical resolution and selectivity of the separation.
- Robustness Module: This module tests the tolerance limits of selected working points by computing the number of Out-of-Specification-(OoS)-results that might occur by given small fluctuations in method parameters, evaluating variable conditions, such as gradient time, temperature, pH or ternary composition, flow rate and start/end points of a gradient.
- DryLab Knowledge Management Module: This is the reporting tool for documenting and archiving a method. It encourages a pharmaceutical Quality by Design (QbD) approach to method development to comply with ICH guidelines for validation as well as USP method validation and FDA bioanalytical method validation standards, ensuring that the process conforms to method validation guidelines by providing a comprehensive method report, including a platform for the step-by-step justification of method choices, with automatically generated Analytical Method Summary for sign off. This assists a laboratory to achieve excellent GMP compliance, along with easier and more effective collaboration between departments, supporting analytical method transfer during development and manufactu-ring.
Resources
Click ICH guidelines for analytical method validation PDF to download detailed requirements.
Click on Ebastine KMD to see a published example of DryLab-generated Technical Specification Document to meet ICH analytical method validation acceptance criteria.
Click on DryLab simulation to see video.