By PHC Europe…
PHCbi COVID-19 vaccine storage solutions
The emergence of a new generation of micro RNA genetic vaccines to combat the global COVID-19 coronavirus pandemic poses new ultra-low temperature storage and cold chain transport challenges for healthcare providers around the world.
PHCEU is able to help meet these challenges with its range of PHCbi’ ultra-low temperature (ULT) biomedical freezers and pharmaceutical refrigerators. These cover the entire range of the cold chain spectrum and the multiple temperature storage points required between manufacture and administration at wholesalers, distribution depots, hospitals, pharmacies and clinics.
mRNA vaccine cold chain challenge
All vaccines are fragile and temperature sensitive. Indeed, the World Health Organization (WHO) has estimated that about half of the vaccines distributed worldwide are wasted, largely due to failures to control storage temperatures properly.
This inherent vulnerability becomes even more serious when it comes to the emerging vaccines being developed to combat the COVID-19 coronavirus. The first candidates to release results from Phase 3 clinical trials, from Pfizer/BioNTech and Moderna, claim effectiveness of up to 95 per cent, an astonishing result. This has been achieved using innovative mRNA technology that targets the ‘spike’ proteins, based on the genetics of the SARS-CoV-2 virus that causes Covid-19, rather than the conventional approach of using the structure or pieces of the virus itself.
However, these fragments of DNA and RNA are very delicate and are liable to degrade quickly on their own unless deep frozen at temperatures as low as -80oC to keep them intact. Once unfrozen, these vaccines also require refrigeration up to the moment of injection.
This poses an unprecedented cold chain and storage logistical challenge with billions of people worldwide to be immunized and each patient requiring two doses, several weeks apart.
TwinGuard secure ultra-low temperature freezers
PHCbi brand biomedical freezers are designed for secure ULT storage of high-value vaccines and other biologics, being based on high performance refrigeration platforms and cabinets engineered for reliability, temperature uniformity, fast temperature recovery, easy control and access and tolerance for real-world conditions.
PHCbi pharmaceutical refrigerators and freezers satisfy any storage protocol or space requirement. Robust refrigeration systems and cabinet designs assure temperature uniformity, reliability and energy efficiency. All are engineered to maintain required temperatures for product viability and to achieve rapid temperature recovery after multiple door openings. The TwinGuard dual refrigeration system provides ultimate temperature security.
These ULT solutions, covering the temperature range from 60 °C to -86 °C , are ideal for storage of mRNA, viral vector and non-replicating viral vector vaccines, as well as specimens.
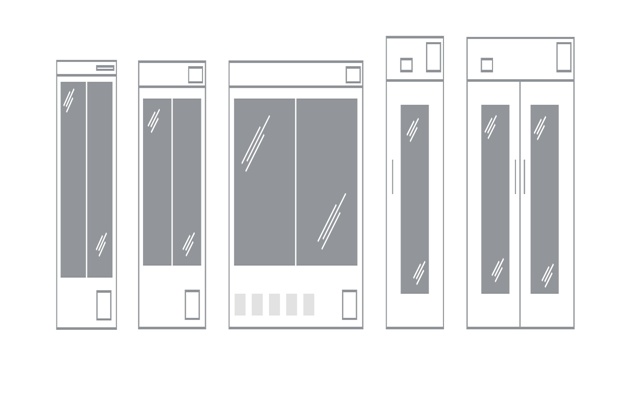
PHCbi’s MDF range of ULT biomedical freezer extends from compact 84 litre top-loading cabinets to high capacity 715L chest storage or 729L single door cabinets (see Resources).
Vaccine Development Freezers
Freezers that cover the temperature range from- 20 °C down to -40 °C are useful in mRNA vaccine development processes for storage of raw materials and some biopharmaceutical ingredients or biological samples.
Here, too, PHCbi meets the challenge with a range of biomedical freezers, including the 479 litre MDF-MU549DH-PE that provides stable temperatures between -20°C and -40 °C , as well as other freezers covering the -20°C to -30°C range and varying in capacity from 369L to 690L (see Resources).
These freezers may also be useful for storage of subsequent vaccines such as the adenovirus vector vaccines being developed by Oxford University/AstraZeneca and Janssen and the Novavax protein adjuvant vaccine indented to boost the immune system. All of these are in various stages of Phase III clinical trials and should be ready for market during the first half of 2021.
Biomedical Refrigerator / Freezer Combos
COVID-19 vaccine programs have highlighted a further need to combine freezing and refrigeration capabilities at point of care, allowing long to medium term freezing and shorter term cool chain storage ready for administration.
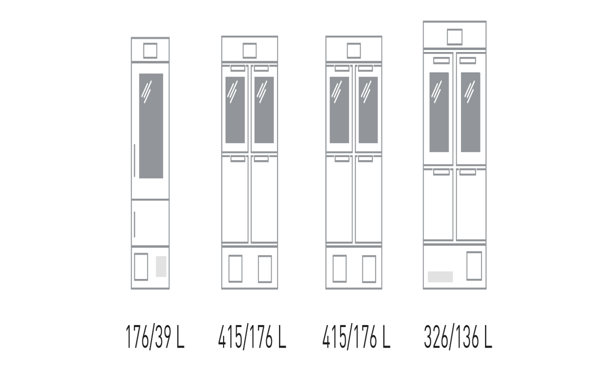
In these situations, PHCEU offer a range of useful solutions in the shape of a wide range of PHCbi biomedical freezer/refrigerator combined units using independent refrigeration systems to provide both -20°C freezing and refrigeration in the +2°C to +8°C temperature range. These combo refrigerator/freezer units include the MPR-215F-PE with capacities of 39L (freezer) and 176L (refrigerator), as well as the larger MPR-N450 series (176L/415L) and the MPR-715F-PE that provides 326L of refrigerated capacity and 136L in freeze down to -30°C. (See Resources).
Pharmaceutical Refrigerators
It is envisaged that even the most temperature sensitive mRNA vaccines against COVID-19 will be able to be kept under cool chain refrigeration (2° C to 8 °C) for a few days in clinical settings immediately prior to administration. For this application, PHCbi offers a wide range of Pharmaceutical Refrigerators ranging in size from 84L up to 845L. (See Resources).
Resources
Click on PHCbi ULT Freezers for full details on Ultra-Low Temperature range.
Click on PHCbi Biomedical Freezers for full product details.
Click on PHCbi Pharmaceutical Refrigerator/Freezer combos for range details.
Click on PHCbi Pharmaceutical Refrigerators for further information.