By Molnár-Institute for…
Molnár-Institute DryLab® helps identify new terazosin impurity profiling method
Berlin: – The Molnár-Institute for Applied Chromatography’s proprietary DryLab®4 chromatography modelling software has played a central role in a newly published research study that proposes a radically more effective impurity profiling method for terazosin than laid out by the European Pharmacopoeia, suggesting alternative columns.
The study, now published in the Journal of Pharmaceutical and Biomedical Analysis, was authored by Molnár-Institute associates and partners Dr. Dániel Enesei, Dr. Imre Kapui and Dr. Róbert Kormány of Egis Pharmaceuticals Plc., in Budapest, Hungary and Dr. Szabolcs Fekete, of the School of Pharmaceutical Sciences, University of Geneva, Switzerland.
This new method is also published as part of the new Ph. Eur. monograph proposal of terazosin in Pharmeuropa issue 32.2.
Faster LC method
This work was motivated by the demand of European Directorate for the Quality of Medicines and Health Care (EDQM) for a new liquid chromatographic (LC) method to be developed for terazosin impurity profiling to replace the old European Pharmacopoeia (Ph. Eur.) method, with the aim of reducing analysis time from 90 minutes.
The Ph. Eur. monograph method is based on two different chromatographic separations to analyze the specified impurities of terazosin. Separate sessions were specified because two of the impurities are not sufficiently retained in reversed phase (RP) conditions, even using 100% water as an eluent. Therefore, next to RP, an ion-pair (IP) chromatographic method had to be applied to analyze those two impurities.
Leading edge hardware and software
The study team used three different liquid chromatographic systems; an Acquity ultra high performance liquid chromatography (UHPLC I-Class) system and an Acquity UHPLC H-Class system both from Waters Corp., and a 1290 Infinity II UHPLC system from Agilent Technologies. The systems were variously equipped with binary or quaternary solvent delivery pumps, auto samplers with flow-through-needle (FTN) sample injectors, column thermostats, and photo diode array detectors.
All chromatographic data were acquired and processed by Waters Empower3 software. UHPLC method development and modeling was performed using DryLab® 4, v.4.3.1 software from Molnár-Institute.
Time reduced from 90 to 20 minutes
The team’s new proposed method makes it possible to profile terazosin impurities in one run in around 20 minutes, less than a quarter of the previous time demanded. It allows increased retention of the two critical compounds using alternative stationary phases, instead of the C18 phase suggested by the old Ph. Eur. method. Applying a pentafluoro-phenyl (PFP) stationary phase made it possible to separate and adequately retain all the impurities. The detection wavelength was also changed compared to the Ph. Eur. method and is now appropriate for the detection and quantification of all impurities using perchloric acid in the mobile phase at low pH.
New workflow proposed
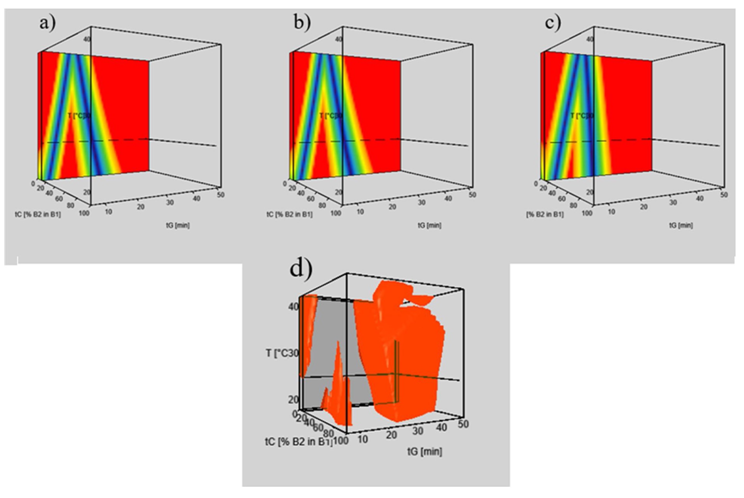
The team also developed a new generic workflow, evaluating chromatographic resolution in a wide range of method variables and have suggested replacement columns for terazosin impurity profiling. They applied retention modeling to study the chromatographic behavior of the compounds of interest and to visualize resolution for the different columns, where given criteria are fulfilled. This allows zone (set of chromatographic conditions) of a robust space to be quickly identified by overlaying individual response surfaces (resolution maps).
The study has also demonstrated that two columns from different providers (Kinetex F5 and SpeedCore PFP) can be used as replacement columns, providing sufficient resolution at the same working point and a high degree of robustness.
About Molnar-Institute
Founded in 1981, Molnár-Institute develops DryLab®, a software for (U)HPLC modeling for a world-wide market. Its powerful modules allow for the most sophisticated method development as required across pharma industries. Analytical scientists use DryLab® to understand chromatographic interactions, reduce runtimes, increase robustness, and conform to Analytical Quality by Design (AQbD) standards.
The Molnár-Institute is a registered vendor to the US FDA, CDC and many other regulatory bodies. DryLab® has pioneered AQbD long before regulatory agencies across the world encouraged such submissions. Widely implemented by thought leaders, the software contributes substantially to the paradigm shift towards a science- and risk driven perspective on HPLC Quality Control and -Assurance.
DryLab®. Right First Time. Every Time.
Resources
Click on Updating the European Pharmacopoeia impurity profiling method for terazosin and suggesting alternative columns to access the full paper.