By Charles Ischi…
Charles Ischi Ph21 Data Logger and the OPC-Server
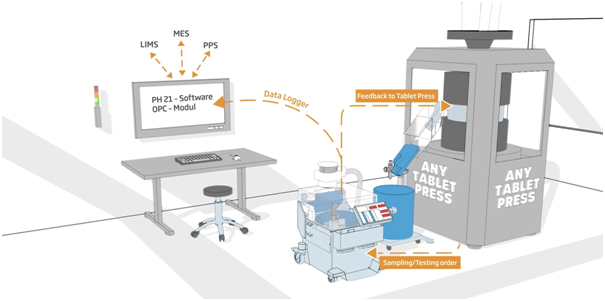
Charles Ischi has developed its innovative Data Logger to capture the binary data stream from tablet testing device to a tablet press. The ‘raw’ data can then be exported into proprietary Ph21 SCADA software to be used for quality control, in-process control, documentation and archiving.
The Data Logger is just one of a wide range of ways of using the OPC data exchange capabilities of the Ischi Ph21 SCADA platform.
Listening to the tablet tester
There are a wide range of tablet testing devices suitable for plugging directly into a tablet press, to be directly controlled by the press.
The Ischi Data Logger monitors the data communication of the test device sending measured values to the press. It captures the data for Ph21 import and analysis, to be used for quality control purposes (e.g. printing test and batch reports).
Ph21 SCADA
The Data Logger is one of a range of add-ons offered by Charles Ischi to expand the functionality of its Ph21 – OPC server-based system, used as a Supervisory Control And Data Acquisition (SCADA) control system architecture comprising computer, networked data communications and graphical user interfaces (GUI) for high-level process supervisory management.
The OPC Data exchange uses the well-established Open Platform Communications (OPC) standard to enable synchronization and structuring of data being exchanged between a PH21 SCADA system and:
- PPC (Production Planning & Control)
- MES (Manufacturing Execution System)
- LIMS (Laboratory Information Management System)
The implementation of such data exchange can be established between Ph21 and Enterprise Resource Planning (ERP) systems, such as Werum PAS-X.
The following tablet test systems running under Ph21 software control can exchange data via OPC:
- Kraemer CIW automatic tablet & capsule weighing systems
- Kraemer UTS universal tablet testing systems for acquisition of weight, thickness, diameter and hardness of solid dosage forms
- Fette Checkmaster tablet testers
- Kraemer HC6.2 manual tablet hardness testers
- Charles Ischi DISI tablet disintegration testers
There are numerous interfaces available for communication and data exchange with external software application and it is also possible to interchange manually acquired values within a Ph21 virtual test environment.
Ph21 software
The Ph21 pharmaceutical quality assurance system enables users to control and evaluate their tablet testers, disintegration testers and weighing machines centrally. Once product-specific data is stored in the central product database, they can be used for all tests on any connected device.
The system supports the connection of external pharmaceutical testing devices, including Kraemer Elektronik’s well-known UTS tablet testing systems, tablet hardness testers and disintegration testers, scales, as well as weighing systems for in-process control.
Full Ph21 software functionality includes production features for full scalability, such as:
- Product library with unlimited number of product specifications
- Out-of-specification Alarms for IPC
- Testing for different shapes and sizes of tablets
- Report creation for individual tests, group of tests, complete batches and annual multi-batch reports
- Menu-guided adjustment and calibration, including reports and calibration interval control
- Multi-tester applications for weight, thickness, diameter, hardness, disintegration, friability etc.
- Direct online testing from tablet press
- Online quality control charts
- Automatic data export functions
- ODT (online data transfer) options for multi-tester applications, managing product and test data from central administrative system, automatically distributed to all testing systems
More than 1,000 Ph21 systems are now in use throughout the pharmaceutical industry worldwide.
Ph21 compliance
The entire software platform and system architecture are 100% compliant with FDA 21 CFR Part 11 regulatory guidelines that define the criteria under which electronic records and electronic signatures (ERES) are considered trustworthy, reliable, and equivalent to paper records for pharmaceutical and healthcare purposes.
This compliance allows users a much broader scope of evaluation options for completed tests, with automatic background backups guaranteeing failsafe in-process control (IPC).
Beginning with the system design, special care has been taken to ensure proper operation of the system. Each new version is released after passing internal testing / verification and validation procedures.
The Ph21 platform also complies with European Regulations for software applications in GMP-critical environments within the pharmaceutical industry.
Resources
Click on OPC-Server Add-ons for further information
Click on Ph21 Software for details on Ph21 Data Acquisition and Analysis