By Charles Ischi…
Charles Ischi solutions for data quality and security in tableting
The increasing regulatory focus on data quality assurance and data security increases the importance of tested and proven solutions that assure data quality control in basic tablet testing and analysis processes.
Charles Ischi Testing Technology offers the Ph21 laboratory automation software that incorporates advanced automated features and meets all main requirements and latest revisions of the US Food & Drug Administration’s (FDA’s) 21 CFR part 11 regulations on electronic records and electronic signatures (ERES).
Use of the Ph21 package means that essential data quality assurance, data integrity, and ERES compliance is built into every part of the data acquisition and analysis involved in tablet testing and oral solid dose (OSD) development.
What is Ph21?
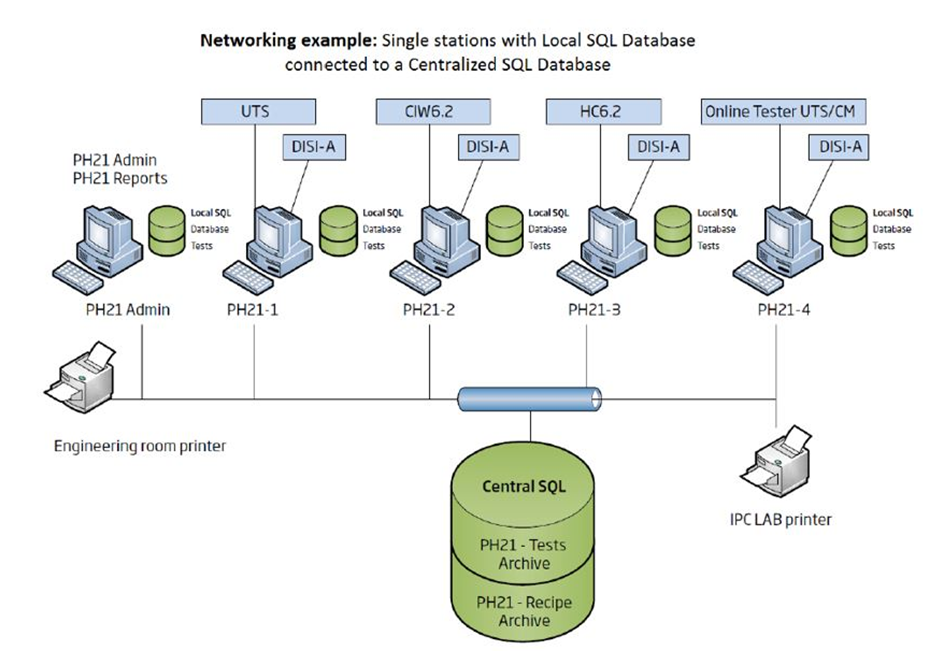
The Ph21 pharmaceutical manufacturing software package is a data quality assurance system that gives users central data quality control from across a wide range of tablet or disintegration testers and weighing machines. Once stored on a central product database, the program allows product-specific data to be applied to all tests on the connected devices.
It uses the well-established Open Platform Communications (OPC) standard to enable synchronization and structuring of data being exchanged between a Ph21 Supervisory Control and Data Acquisition (SCADA) control system and a PPC (Production Planning & Control) process development system.
The system supports the connection of up to 32 external pharmaceutical testing devices, including Kraemer Elektronik’s well-known UTS tablet testing systems, tablet hardness testers, Ischi’s disintegration and friability testers, scales, as well as weighing systems for in-process control. For even larger applications, the Ph21 system can be installed and operated as a client-server application with numerous interfaces available for communication and data exchange with external software applications, allowing flexible data export to in-house Data Management System (LIMS, OPC, MES).
FDA 21 CFR part 11 requirements
The entire Ph21 software package is 100% compliant with the FDA’s 21 CFR Part 11 requirements that define the criteria under which electronic records and electronic signatures are considered trustworthy, reliable, and equivalent to paper records.
This built-in compliance allows users to choose a wider scope of evaluation options for completed tests. Automatic backups in the background guarantee failsafe in-process control (IPC).
Examples of Ph21’s integral data controls include:
- Automatic data completeness check whenever an event is stored with incomplete entries blocked.
- All relevant activities automatically logged in Ph21’s Audit Trail with date/time, name and responsible person ID, as well as unique identity of device involved.
- All activities and changes protected by password Log In and assigned authorities
- Change control system within Ph21 Audit Trail backed up by Master Data change control log.
- Data Confirmation feature allowing authorized second person to review data and confirm record, making them protected from future alterations.
- Comprehensive ‘Archive’ feature that automates archiving and further protects data integrity from subsequent alteration.
The entire software platform and system architecture are thus 100% compliant with FDA 21 CFR Part 11 regulatory guidelines that define the criteria under which electronic records and electronic signatures (ERES) are considered trustworthy, reliable, and equivalent to paper records for pharmaceutical manufacturing and healthcare purposes.
In the core system design, special care has been taken to ensure proper operation of the system, with each new version only being released after passing the internal testing / verification and validation procedure.
The Ph21 platform also complies with European Regulations for software applications in GMP-critical environments within the pharmaceutical industry.
Resources
Click on Ph21 Software for details on Ph21 Data Acquisition and Analysis.