By Charles Ischi…
Charles Ischi CI-AG Tablet batch analysis
Ensuring and maintaining Quality Assurance (QA), Quality Control (QC) and Good Manufacturing Practice (GMP) standards in tablet testing and manufacturing depends on batch testing products.
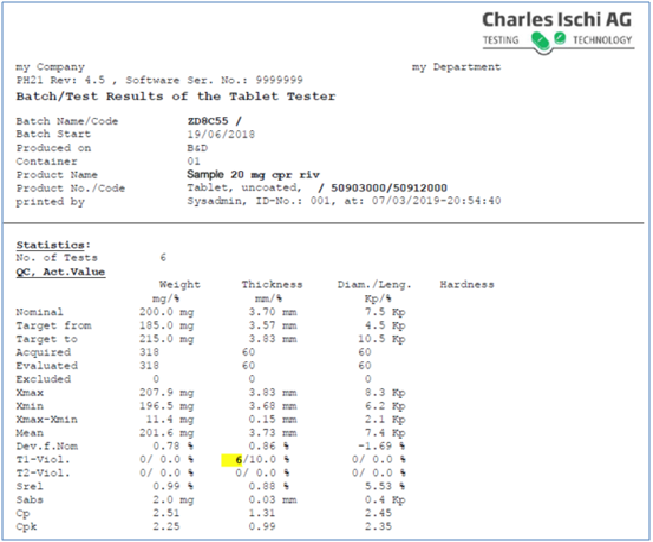
These batch proof reports will include key parameters such as API purity, residual solvents or heavy metals, particle size, bacterial or microbial limits as well as tablet properties such as weight variability, hardness, friability, disintegration or dissolution performance . Batch records therefore form key reporting elements for both regulatory approval and inspection and thus must be kept in secure formats that are also quickly accessible and capable of quick and easy analysis for ongoing QC.
Charles Ischi AG (CI-AG) Testing Technology offers its Ph21 laboratory automation software and add on Ph21 Reporting Tool as a comprehensive solution to the challenges of maintaining and accessing electronic batch records.
What is a batch?
A batch is normally defined as any quantity or consignment of goods produced at the same time and under the same conditions (e.g. using the same formulation and/or the same machine).
The master batch record will record the results from every batch produced during testing, from Batch 1 onwards and will include batch formula records, batch manufacturing records, and batch production records.
Batch reporting challenges
Enterprise resource planning (ERP/MES) tools, covering operational end-to-end manufacturing, supply and other processes, have often been used to maintain basic records of batch statistics. However, use of ERP systems as electronic batch record systems have at least three potential shortcomings when it comes to meeting regulatory batch record keeping expectations, as well as the technical requirements of 21CFR Part 11. These are:
- Assuring batch record data integrity.
- Formatting batch records to meet regulatory requirements.
- Including all of necessary content in the batch statistics, including attachments.
These issues are to some extent interlinked and overlapping and can only be fully addressed to meet the full expectations of regulatory authorities by considering all elements holistically as part of a fully integrated electronic batch record software solution. The CI-AG Ph21 software package and its Ph21 Reporting add on provides fast access to batch data archives and the ability to quickly isolate specific batch record data regarding the physical parameters of previously tested tablets.
Ph21 software platform
Charles Ischi’s Ph21 pharmaceutical quality assurance system provides users with centralized control of tablet testing systems, allowing them to integrate individual testing stations and processes into a whole system. Once product-specific data is stored in the central database, it is available throughout the system.
For larger applications, the Ph21 system can be installed and operated as a client-server application. The entire software is 100% compliant with FDA 21 CFR Part 11 as well as the European Regulations for software applications in GMP-critical pharma environments and allows a wide scope of evaluation options for completed tests. Automatic backups in the background guarantee failsafe in-process control (IPC).
Numerous interfaces are available for communication and data exchange with external software applications.
Features from the full version of the Ph21 software include abilities create reports of individual tests, group of tests, complete batches and multi-batch reports (annual reports), automatic data export functions, and ODT online data transfer options for multi-tester applications, where product and test data may be managed from a central administrative system and automatically distributed to all testing systems.
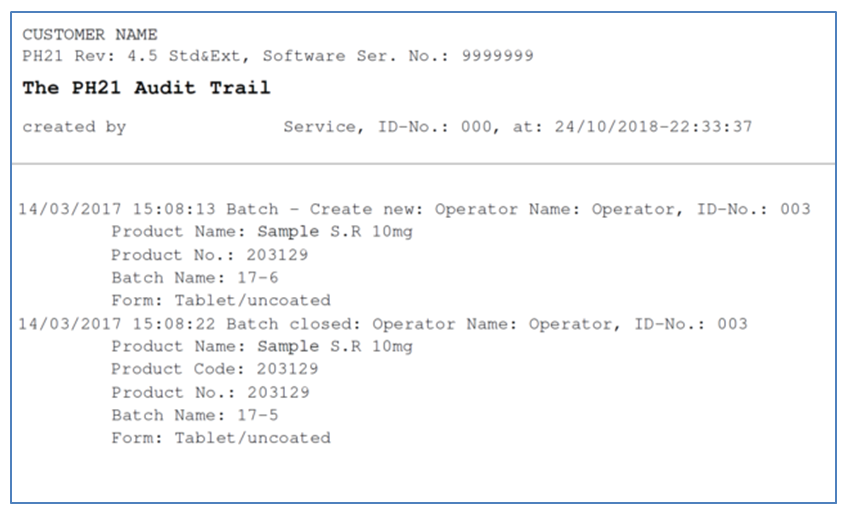
The Ph21 system is designed for robust and reliable quality and in-process control in the laboratory as well as for production lights-off operation. Automatic sampling, testing and generating of test protocols, including online quality control charts with SPC-functions (Statistical Process Control) makes it state-of-the-art software for batch reporting, with all significant events logged for later retrieval and analysis. The Auto-Archive function automates saving of batch data direct to SQL database server, where the data are backed up and saved.
Ph21 Reporting Tool
The Reporting Tool is particularly useful in retrieving meaningful and compliant test data on specified batches from historic unstructured archives for Audit purposes. The software can access batch archives directly, making it unnecessary to restore or adapt them to new formats, thus preserving data integrity.
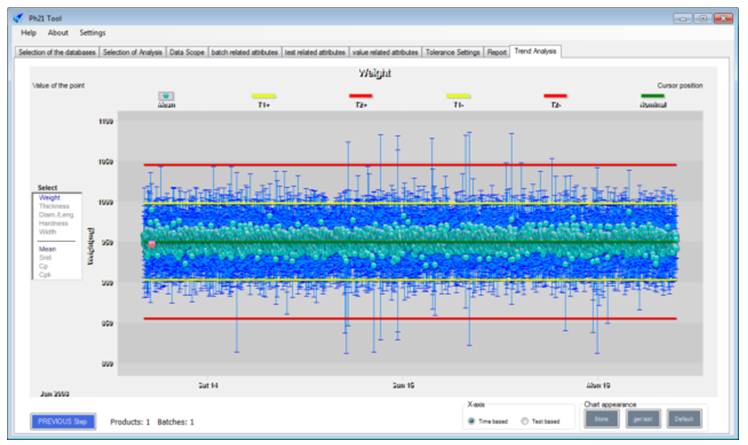
The tool can also generate sophisticated graphical representative package diagrams, allowing huge quantities of data to be searched in very intuitive ways. This allows batch data from several Ph21 archives to be summarized and analyzed without needing to retrieve the data from the archive.
It can also quickly identify tolerance violations, using sophisticated analysis methods such as CSFT/DT, or audit trail analysis/searching, etc., to filter out implausible values that are harder to detect than simple wild codes because they normally require insights or recall on what was being measured, making cleaning implausibles from very large data sets very time and labour-intensive.
The Charles Ischi Ph21 Reporting Tool allows the user to conduct specific searches for implausibles, filtering searches by product name, number or code, as well as batch names or codes. It also supports specific ‘find’ functions where user enters an exact or partial expression that the data must contain.
The Reporting Tool allows user to generate reports, either as Overview, containing individual measurements with implausibles, or Retailed Report containing full tests, including some implausibles. It also allows data analyses to be exported to MS ExCel for further processing.
The external Reporting Tool can process archive data more than ten times faster than the standard reporting feature implemented within the Ph21 package.
However, because data cleaning is such a processor intensive task, it has been designed to run as a stand-alone package, being hosted on server or a separate PC, to relieve processing pressure on the core Ph21 test system.
Resources
Click on Charles Ischi Ph21 Software for Data Acquisition and Analysis for more information.
Click on Charles Ischi Ph21 Reporting Tool for more information.