By Bachem AG
Bachem GMP peptide and oligonucleotide manufacturing
Bachem acquired early mastery of development and manufacturing of peptides, the short chain amino acids linked by peptide bonds that have enabled new generations of small molecule drugs that closely mimic the body’s natural pathways. Now it is extending that expertise to oligonucleotides, the short single strands of synthetic DNA or RNA that serve as building blocks for many molecular biology therapies.
Bachem possesses world-class capabilities to manufacture peptides and oligonucleotides at industrial scale and in full compliance with the strictest Good Manufacturing Practice (cGMP) standards.
Increased demand for oligonucleotides
Peptides continue to enjoy prominence in mainstream pharmaceutical applications, particularly in ‘replacement therapies’, with more than 60 peptides currently approved by North American, European and Japanese health regulators and more than 150 others in development.
Meanwhile oligonucleotides have matured into a drug class with a broad indication spectrum and are emerging as new treatment options in rare and orphan disease areas.
The promise of oligonucleotides making a difference to an increasing number of patients is driving Bachem’s rapid implementation of manufacturing capabilities and capacities for oligonucleotide-based active pharmaceutical ingredients (APIs).
Bachem oligonucleotide development
With decades of experience in the development, production and regulatory support of APIs, Bachem is extending its focus from peptides to oligonucleotides with the aim of becoming the first-choice oligonucleotide manufacturer. Since the technologies are fundamentally different, this has involved meticulous transformation of the organization with strong focus on innovation and quality.
Nevertheless there are synergies between peptide and oligonucleotide manufacturing since both require expert knowledge of solid-phase synthesis, protecting group chemistry, purification by chromatography, ultra/diafiltration techniques, precipitation and lyophilization. Bachem has therefore been able to tap into its accumulated peptide knowledge base in establishing itself as a world-class Contract Manufacturing Organization (CMO) for oligonucleotides.
Oligonucleotide cGMP manufacturing capabilities
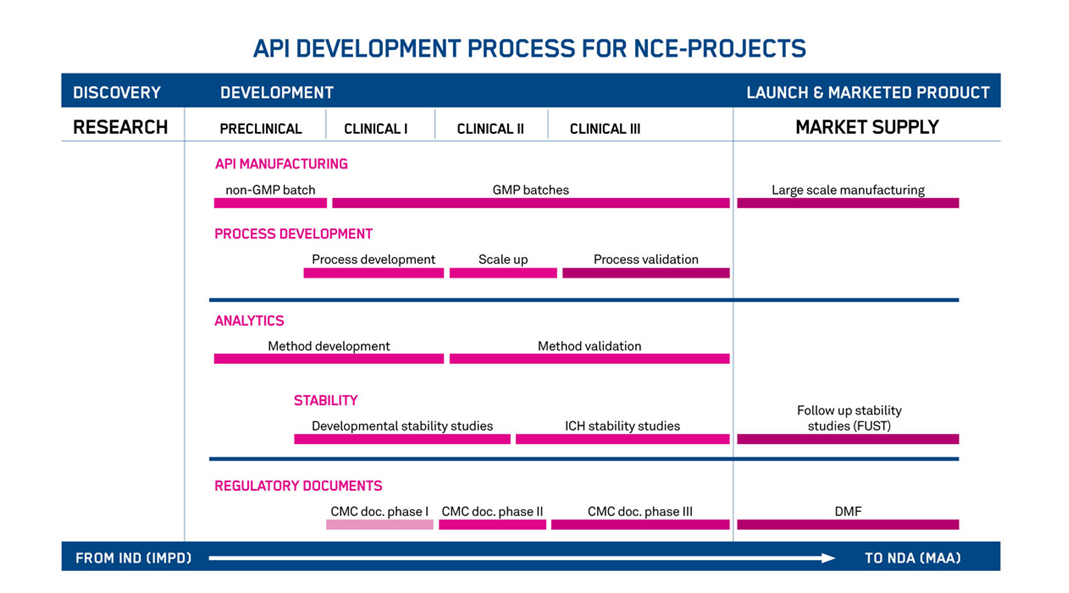
Bachem offers a full range of Chemistry, Manufacturing and Control CMC development services for oligonucleotide-based APIs starting with the establishment of scalable manufacturing processes and ending with IND and NDA applications supported by Bachem’s in-house regulatory affairs specialists.
Bachem brings innovation to nearly every piece of equipment and process from the oligonucleotide synthesis to the isolation of the API. Bachem offers process development and GMP manufacturing services for synthetically accessible oligonucleotides at multi–kilo scale and has designed an equipment train with new engineering solutions for tight process control and high throughput.
These new chemistry and manufacturing capabilities are complemented by the support of Bachem’s exceptional analytical chemistry team and their long-standing expertise in developing and validating test methods for large and structurally complicated molecules.
Bachem’s facilities are fully certificated for GMP compliance, being regularly inspected and approved by national and international regulatory authorities like FDA, Swissmedic and others (see Resources).
A clear plan for developing the CMC management of peptide and oligonucleotide-based drug substances offers Bachem’s partners a risk-mitigated approach to any clinical and commercial milestones.
Bachem production plants are endowed with state-of-the-art equipment for solvent supply, peptide and oligonucleotide synthesis, purification and isolation of active ingredients and intermediates. All equipment and containment is GMP qualified and cleaning validated. Overlapping capacities and sizes of different equipment trains facilitate a smooth scale-up for increases in demand within the product life cycle.
Upstream peptide and oligonucleotide synthesis
Bachem production plants are equipped with solution-phase synthesis reactors as well as solid-phase synthesis reactors. Facility design makes it possible to run combinations of the two techniques in a hybrid approach. Thy are also equipped to run modern Green Chemistry approaches such as Molecular Hiving™.
For a wide range of solution phase chemistries, Bachem utilize reactors up to 8,000 liters. For solid phase peptide synthesis (SPPS), it uses reactors up to 1,000 liters, with larger scale-up options currently under construction. In Bubendorf, a new large-scale facility is being planned to handle peptide liquids and oligonucleotide with reactor sizes up to 3,000 liters.
Bachem maintains multiple independent production trains for each type of GMP qualified equipment for oligonucleotide synthesis, Bachem uses cGMP pharmaceutical-qualified OligoPilot™ 100 and 400 synthesizers plus a large-scale synthesizer, currently under construction, with up to 2 mole capacity. Custom-made cleavage and deprotection equipment trains enables wide range of chemistries at all scales. Handling of large quantities of hazardous reagents is achieved in a closed and safe environment.
Bachem balances the flexibility of key equipment, with various levels of automation, with increasing use of a Manufacturing Execution System (MES) and a process data historian. Processes are developed according the Quality by Design (QbD) approach, controlled via in-process controls (IPC) or process analytical technology (PAT).
Downstream purification and isolation of peptides and oligonucleotides
Bachem is committed to the systematic expansion and modernization of its purification equipment in order to ensure the efficient production of ever-increasing amounts of bulk peptide and oligonucleotide pharmaceuticals.
It uses sophisticated methods for large scale purification campaigns such as preparative high performance liquid chromatography (HPLC), ion exchange (IEX), size-exclusion chromatography (SEC), and ultra-filtration (UF/TFF). The equipment in place permits highly efficient or even continuous manufacturing of extremely pure products up to multi-kg quantities per lot.
For preparative HPLC, dynamic axial compression (DAC) stainless steel columns of up to 60 cm diameter both in batch and continuous mode are packed with the appropriate high performance silica separation phase. For low-pressure chromatography columns up to 80 cm diameter are available. Solvent delivery is ensured from eluent tank farms and containers.
The control of microbiological contamination is a prerequisite for API manufacturing. Class D (ISO 8) and C (ISO 7) clean rooms are supplied via HEPA-filtered, temperature and humidity controlled air, down-flow booths are used for minimizing microbial contamination and protecting operators. Highly active pharmaceutical ingredients are handled in integrated safety workbenches or flexible isolators reaching OEB level 4 (1-10 µg/m3).
Predefined physicochemical properties of the API can only be achieved by a carefully controlled isolation process. Besides precipitation and crystallization, lyophilization of intermediates and final API is a standard unit operation. On each site Bachem have multiple lyophilizers in different sizes (up to 300 liters) located in clean rooms.
Small molecule manufacturing
Bachem’s capabilities for the cGMP pharma production of GMP small molecules are concentrated at its site at Vionnaz, Switzerland, which offers a range of specialized services .
Small molecule production capabilities include: process development, chiral synthesis, heterocyclic chemistry, metal-catalyzed reactions, hydrogenations, oxidations and reductions using various reagents, enzymatic reactions, and high pressure reactions.
Resources
Click on Perfect Ingredients for a Better World to view video.
Click on Bachem cGMP production to learn more.
Click on Bachem Therapeutic Oligonucleotides for scientific information.
Click on Bachem products to browse Bachem online shop.
Click on Bachem certification to see copies of cGMP certificates, licenses and other documents.
Click on Bachems News to see latest News & Events.