By Dec Group
Air Pollution, DPI and Micronization
Materials handling and containment solution specialist Dec Group has developed a new generation of spiral jet mills meeting more demanding particle size distribution (PSD) targets for dry powder inhalers (DPI).
At the same time, in the face of ever-increasing levels of chronic obstructive pulmonary disease (COPD), Dec recognizes the need for safe handling and processing of highly potent active pharmaceutical ingredients (HPAPIs), creating further micronization and powder handling challenges in developing new generations of DPI therapies.
Global COPD challenge
The World Health Organization (WHO) recently reported that an estimated 90 per cent of the global population are subject to high levels of air pollution, reflected in rising levels of COPD. On current estimates, some 65 million people worldwide suffer from COPD, responsible for around three million deaths annually, making it the third leading cause of death worldwide.
One of the worst affected countries is the People’s Republic of China, where COPD is widespread with 8.6 percent of the country’s adult population – almost 100 million people – suffering from chronic lung disease.1
According to the Global Initiative for Asthma, there are more than 500 million asthma patients worldwide, with over 30 million in China. This number increases by 4% annually.
Therapies
Most asthma and COPD medications are inhaled, using an inhaler (or nebulizer) to deliver medicine directly to the lungs. Some drugs are also available in tablet, infusion or injectable form.
Asthma inhaler devices include nebulizers, metered dose inhalers (MDIs), breath actuated inhalers soft mist inhalers (SMIs). However, the most important class of inhalation devices are Dry Powder Inhalers (DPIs) that have many unique advantages that have contributed to the incredible growth of DPI pharmaceutical products. A relatively large number of DPI devices for various inhalable powder formulations are available to increase performance.
DPI and Micronization
The Aerosol Society (aerosol-soc.com) has carried out a study that shows that the inhalation performance of a DPI is strictly correlated with the critical quality attributes (CQA) of the active pharmaceutical ingredient (API).2
Particles typically need to be reduced in size to within the aerodynamic diameter range of 1-5 µm in order to reach the airways. These dimensions are usually targeted when micronizing crystalline APIs.
For this purpose, jet mills are a leading micronization technology, achieving precise particle size reduction by using air or nitrogen at very high pressure.
Advanced micronization technology
Achieving high performance with conventional jet mills remains very difficult. The two most important critical process parameters (CPP) which directly affect the API particle size are micronizing pressure and feed rate. Nowadays it is of fundamental importance for a manufacturer to rely on a technology able to achieve the desired DPI inhalation performance through direct control of the micronizing parameters.
Therefore, Dec has set itself the task, in close cooperation with global pharmaceutical partners, of creating advanced spiral jet mills that can meet the most demanding particle size distribution (PSD) targets for dry powder inhalers (DPI).
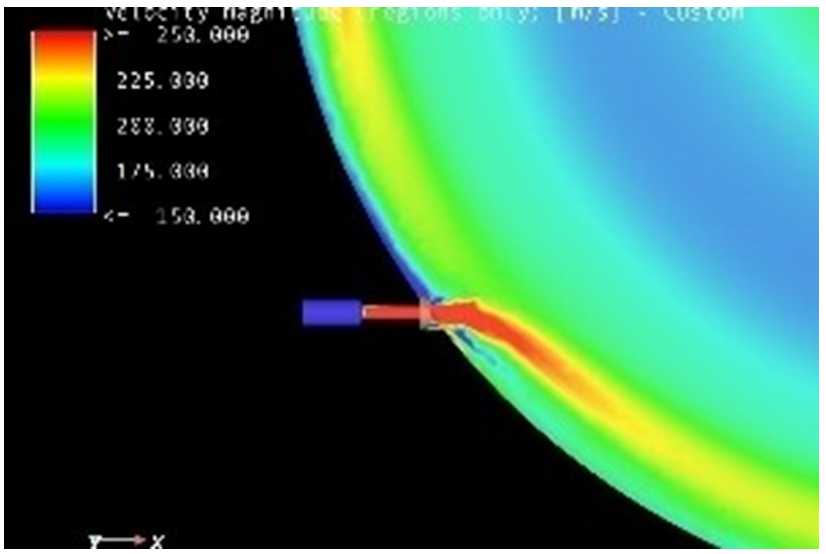
A primary aid in this development has been the use of Computational Fluid Dynamic (CFD) modelling, allowing mill performance to be studied dynamically across a range of operating parameters.
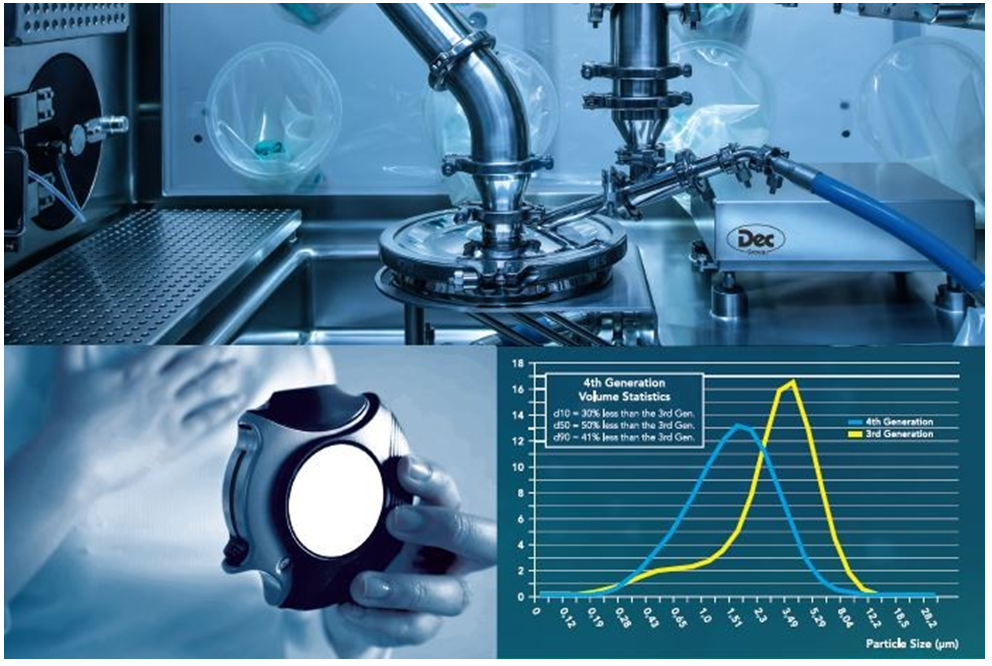
CFD modelling is reflected in the design of Dec’s patented 4th generation MC DecJet® range of spiral jet mills that are able to micronize corticosteroids such as fluticasone while maintaining a very tight PSD curve with a typical measurement of d50 < 2 µm, representing a 50% improvement in terms of cumulative distribution and impeccable scalability from research to production models. The carved chamber MC DecJet® version with low-friction inserts is also the product of CFD-assisted research.
The end-user benefit is more effective drug deposition deep into the small peripheral airways of the lungs.
HPAPI focus
The MC DecJet® micronizing and containment range (from MC DecJet® 30 to MC DecJet® 400) are products of a perfect union of years of experience as user and provider of micronizing and containment technologies, with the latter also becoming ever more relevant.
The global interest in developing improved therapies for pulmonary and other diseases has also led to increased focus on use of highly potent active pharmaceutical ingredients (HPAPIs). In COPD, higher potency drugs typically have the potential to achieve similar or better efficacy than other drugs at a lower dose.
However, these more powerful substances also require a hazard and risk-based approach in ensuring safe handling methods and high containment. The most common methods for classifying highly potent active ingredients are based on control banding strategies, which categorize APIs based on occupational exposure limits. An occupational exposure limit (OEL) is an upper limit for the acceptable concentration of an active substance in the air at the workplace. Therefore, drug manufacturers not only place extremely high demands on micronization performance, but also on containment strategies and high dosing accuracy.
Isolated micronization
Dec’s micronizing isolators provide a safe enclosure around the micronization process and therefore high containment of potent and/or sterile compounds for both operator and product protection.
Dec can offer highly complex integrated and completely contained solutions from the handling of the incoming powders, to micronization and micro-dosing within the isolator for inhaler filling operations that meet current and emerging needs.
Years of experience and deep-rooted expertise have made Dec a world leader in both micronization and process containment technologies also able to provide technical customer support for their process development and optimization.
With a large number of solutions for particle size reduction implemented all around the world, of which DPI production is becoming increasingly important, Dec is helping to improve life with asthma thanks to its highly contained concepts allowing to achieve the desired DPI inhalation performance through the control of the particle size reduction process parameters.
References:
- Wang C, Xu J, Yang L, Xu Y, Zhang X, Bai C, Kang J, Ran P, Shen H, Wen F, Huang K, Yao W, Sun T, Shan G, Yang T, Lin Y, Wu S, Zhu J, Wang R, Shi Z, Zhao J, Ye X, Song Y, Wang Q, Zhou Y, Ding L, Yang T, Chen Y, Guo Y, Xiao F, Lu Y, Peng X, Zhang B, Xiao D, Chen CS, Wang Z, Zhang H, Bu X, Zhang X, An L, Zhang S, Cao Z, Zhan Q, Yang Y, Cao B, Dai H, Liang L, He J;, Prevalence and risk factors of chronic obstructive pulmonary disease in China: a national cross-sectional study. Lancet. 2018 Apr 28;391(10131):1706-1717. doi: 10.1016/S0140-6736(18)30841-9. Epub 2018 Apr 9. PMID: 29650248.
- Hadrell, A, Directly Probing the Dynamic Behaviour of Particles Originating from DPI and MDI Starting Formulations, Poster, Annual Aerosol Science Conference 2018: https://aerosol-soc.com/abstracts/directly-probing-the-dynamic-behaviour-of-particles-originating-from-dpi-and-mdi-starting-formulations-2/
About Dec Group
Dec (Dietrich Engineering Consultants) Group is a leading global provider of contained powder handling systems and is recognized as a world-leading expert in process containment technologies. It has been supplying the pharmaceutical, chemical, food and cosmetic industries for more than 30 years.
Headquartered near Lausanne, Switzerland, Dec Group has a global presence with subsidiaries and agents active in more than 60 countries including subsidiaries in the UK, Ireland, Germany, Poland, the Netherlands, India, China and the USA.
In excess of 600 companies worldwide have successfully integrated more than 10,000 Dec systems and concepts into their production sites, always enhancing safety, containment and productivity.
Dec offers innovative approaches across the range of powder handling and process containment applications, including transfer, micronizing/milling, bulk handling, sampling, blending, dosing, aseptic fill-finish solutions, process isolators and containment solutions as well as advanced cleaning (CIP/SIP) features.
Resources
Click on Dec Group Testing Facilities to watch video.
Click on Introduction to Dec Group to watch video.