By Bachem AG
Solid Peptide Synthesis
Solid-phase peptide synthesis (SPPS) is the core technique for producing peptides at various scales. Also known as Solid Peptide Syntheses (SPS), this is a crucial technology in which Bachem is a world leader.
SPPS explained
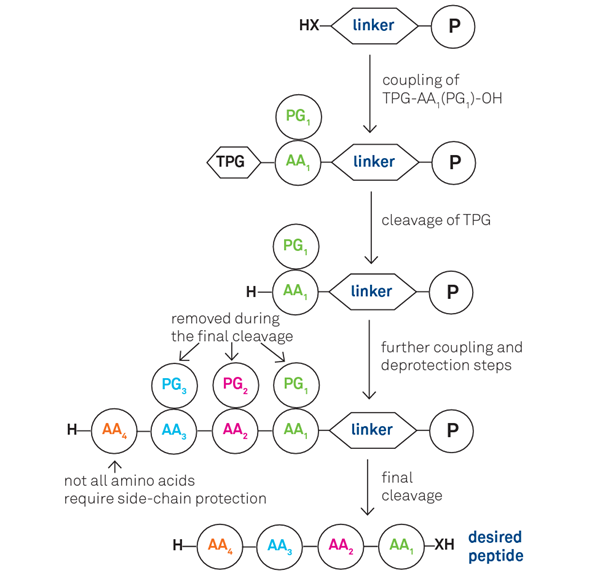
SPPS can be defined as a process in which a peptide anchored by its C-terminus to an insoluble polymer is assembled by the successive addition of the protected amino acids constituting its sequence.
Each amino acid addition is referred to as a four-stage cycle:
- Cleavage of the Nα-protecting group
- Washing steps
- coupling of a protected amino acid
- Final washing
With growing chain bound to an insoluble support, simple filtration is effective in removing excess reagents and soluble by-products. Washing with appropriate solvents ensure the complete removal of cleavage agents after the deprotection step, along with excess reagents and by-products resulting from the coupling step.
Boc or Fmoc?
Once the sequence has been completed, the peptide must be cleaved from the growing resin. Acidolytic cleavage from the resin is generally the method of choice to finally release the peptide. However there has been increased uptake of a broader range of resins susceptible to be cleaved by nucleophiles such as the “Kaiser oxime resin”, along with methods such as p-carboxybenzyl alcohol linker or by photolysis.
The first step in achieving successful synthesis is selection of the right combination of protecting groups with solid support. For standard SPPS this choice is generally limited to a butyloxycarbonyl (Boc) combined with benzyl or a Fluorenylmethyloxycarbonyl Fmoc/tBu based scheme.
Cleavage moieties are often incompatible with the conditions of Fmoc SPPS, whereas allyl-based anchors are resistant towards the cleavage conditions of Boc as well as FMOC protecting groups. The so-called «safety-catch linkers» are perfectly compatible with both Boc and Fmoc chemistries. Only after an activation step are they highly sensitive towards nucleophiles, such as the sulfonamide linker or 4-hydrazinobenzoic acid.
The Boc group was used almost exclusively during the first decade of SPPS, permitting remarkable synthetic achievement. Recently, the introduction of the newer Fmoc protecting group has offered more flexibility for the modification of the peptide chain and/or more specificity in the cleavage of the Nα– versus the side-chain protecting groups.
The combination Fmoc/tBu has met these requirements and broadened the scope of SPPS. Moreover, the development of new resin derivatives has allowed the cleavage of fully protected sequences which can be further coupled in SPPS or in a classical solution process.
In addition, a variety of selectively cleavable protecting groups offers new perspectives for «on-resin» modification (cyclization, formation of disulfide bridges, derivatization of side chains, etc ).
Manual Synthesis
The ‘classic’ SPPS reactor merely consists of a cylindrical vessel with a fritted porous glass disc and a removable lid equipped with a mechanical stirrer. The resin may also be stirred by bubbling nitrogen through, however more elaborate equipment is required. For rapid small-scale synthesis, a small fritted glass funnel is sufficient. Oxygen and moisture need not be strictly excluded, but the cleavage of the Nα protecting group should be performed under a hood as to avoid exposure to piperidine (Fmoc cleavage) or TFA (Boc cleavage).
The swelling of the resin must be taken into consideration in the choice of the reactor size. Normally, the volume of the swollen peptide resin will slowly increase during chain elongation. When synthesizing a medium-sized peptide (20–30 AA) using Fmoc SPPS, a 100–150 ml reactor will suffice for ca. 10 g of resin. The swelling will be more important in Boc SPPS mostly during the TFA deprotection step; a 250 ml reactor would be recommended for the above- mentioned synthesis.
At the beginning of each coupling cycle, deblocking or washing step the resin and the solution must be mixed thoroughly, followed by slow stirring or shaking for the remaining process. All the beads must be suspended in the liquid for thorough washing, efficient coupling, and complete deblocking. It is important to watch for beads sticking to the wall of the vessel especially during the coupling and rinse them from the wall with a small amount of solvent if necessary. Overly vigorous stirring may cause the beads to ‘stick’ to the wall of the vessel. This problem can be overcome by silylation of the glassware to improve surface hydrophobicity.
Solvents are filtered off by slight suction, or, more gently, by applying inert gas pressure. In Fmoc/tBu based SPPS the vessel may also be used for the final cleavage or for the cleavage of fully protected peptides from very acid-labile resins.
Continuous Flow Synthesis
The continuous flow approach packs the solid support into a column, with reagents and solvents delivered by a pump. The resins used in this technique must be able to withstand considerable pressure and, at the same time, keep a constant volume while changing solvents. The standard polystyrene-based resin is not suitable for that purpose as the volume of the beads markedly depends on the solvent. This type of peptide synthesizer is best used for Fmoc-based protocols. The Boc protocols generate ionic species during the Boc cleavage, which cause considerable changes in swelling due to electrostatic forces. A synthesizer has been developed in which swelling is monitored, considering that during Fmoc-SPPS, volume changes in each solvent can only be caused by the growing peptide chain.
Composite material made from a rigid support such as Kieselguhr particles or large pore crosslinked polystyrene in which dimethylacrylamide has been polymerized are used for continuous flow synthesis. Poly(ethylene glycol)-based supports such as TentaGel or PEGA have been introduced for batch as well as continuous flow synthesis.
Fully Automated SPPS
A variety of fully automated synthesizers for batchwise and continuous flow SPPS is commercially available. Fully automated synthesizers employing microwave irradiation for accelerating the synthetic steps were successfully introduced to the market.
Fmoc/tBu SPPS permits automatic monitoring and adequate adjustment of deprotection and coupling times to achieve complete conversions. The monitoring relies on strong chromophores which are either released during deprotection or «consumed» (the Fmoc amino acid derivative) and concomitantly released (HOBt or HOAt) during coupling. Monitoring via changes of conductivity allows the monitoring on a real-time basis and end point value can be given to determine the completion of the coupling reaction.
Resins
All resins at Bachem are obtained from beaded polystyrene crosslinked with 1% divinylbenzene (a mixture of the meta and the para isomer). This degree of crosslinking is optimal for SPPS. A higher level of crosslinking would reduce the swelling whereas a decrease would cause a considerable loss of mechanical stability in the swollen state.
The carrier resins for SPPS are obtained from this polymer or from the chloromethylated material. In the second case, the available load is restricted by the degree of chloromethylation. The average bead size is adjusted by the conditions of polymerization. Bachem offers the most popular size distribution 200–400 mesh (average diameter 38–75 μm). A variety of resin derivatives is also available as large beads: 100–200 mesh (average diameter 75–150 μm). With such resins, reaction times may have to be prolonged due to limited diffusion towards the interior of the beads.
The load of the resins is adapted to the needs of routine SPPS: 0.7–1 mEq/g before the loading of the first Fmoc amino acid. Loads may be deliberately reduced, e.g., for side-chain cyclization, for the synthesis of long peptide chains (above 30–40 residues), or for the preparation of sequences presenting intrinsic difficulties. Resins having a particularly high load can be prepared by Bachem on request.
Linkers
Linkers are bifunctional molecules anchoring the growing peptide to the insoluble carrier. Linkers may be coupled to any carrier suitable for SPPS, an important option where alternatives to polystyrene-based resins are being sought.
The C-terminal Fmoc amino acid may be coupled to the linker yielding the so-called handle which can be purified before loading the polymer. High loads regardless of the bulkiness of the amino acid are obtained by coupling these handles.
Alternative peptide synthesis techniques
Bachem has other techniques available to produce peptides, of which the two most important are:
-
- LPPS: Liquid-Phase Peptide Synthesis has largely been supplanted by SPS for small-scale batch production but is attracting growing support as a industrial-scale peptide synthesis method of choice, since it combines the advantages of Classical Solution Peptide Synthesis (CSPS) and SPS, with peptide elongation carried out in liquid solution and the growing peptide chain is supported on a soluble tag, which confers characteristic properties. LPPS protocols allow the large-scale production of peptides and reduce the use of excess reagents and solvents, supporting ‘green chemistry ‘approaches.
- Molecular Hiving™: This is a highly advanced technique that can be used to produce shorter peptides without hazardous solvents, with more efficient scale-up and enhanced process controls, again as part of a green chemistry strategy.
Resources
Click on Innovative approaches to large-scale peptide production to learn more on Molecular Hiving™.
Click on Innovations for Tides to download brochure.
Click on Bachem 360 Live to watch recent webinars on peptide synthesis (login/sign up required).
Click on Bachems News to see latest News & Events.