By Skyepharma Production…
Skyepharma’s expert CDMO compression of complex tablets
CDMO solutions provider, Skyepharma Production S.A.S. (Skyepharma) has a suite of sophisticated facilities and capability to answer pharma industry needs for compression manufacturing of complex combination for the 3rd and 4th generation oral dosage form (ODF) tablets.
These tablets typically combine several active pharmaceutical ingredients (APIs) in one ODF to increase bioavailability, simplify dosage regimes, or targeting very specific aspects of illness through interaction with particular regions of the GIT (Gastro-Intestinal Tract).
Complex compression for sophisticated dosage release
Manufacturing these complex tablets requires the advanced expertise and technologies needed to undertake multilayer tablet compression and manufacture of press-coated (or tab-in-tab or dual core) ODFs.
To answer these challenges, Skyepharma has a number of advanced solutions to offer its clients, including its own well-proven proprietary Geomatrix® and Geoclock® complex tablet compression technologies. These represent different approaches in meeting the challenges posed by complex dosage release profiles increasingly demanded for effective drugs.
Geomatrix® multilayer tablets
Geomatrix® multi-layer tablet technology is a well-established, validated and customizable oral drug delivery platform technology that is currently used in products now commercialized worldwide, in Europe, USA and Brazil.
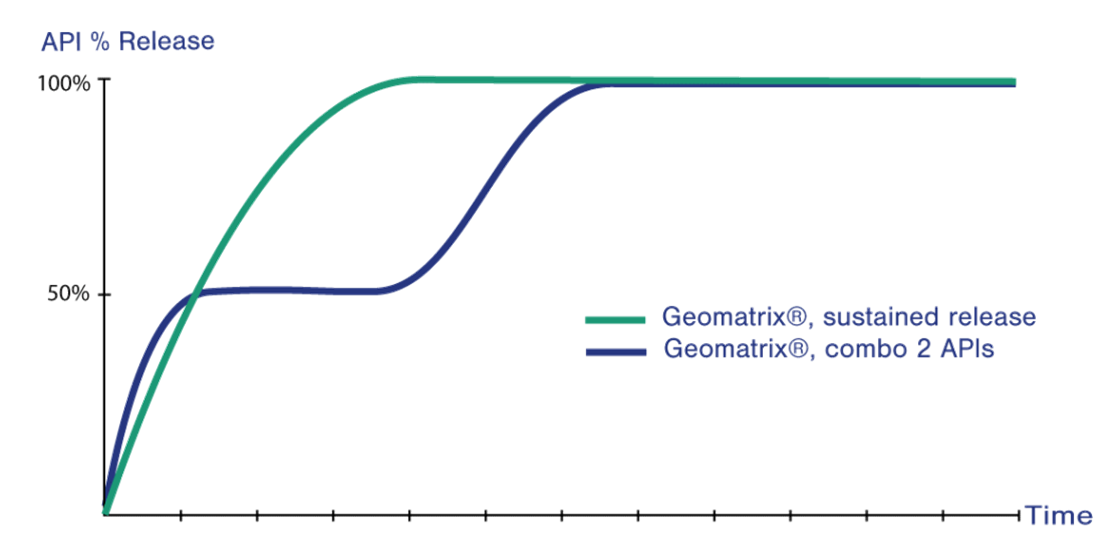
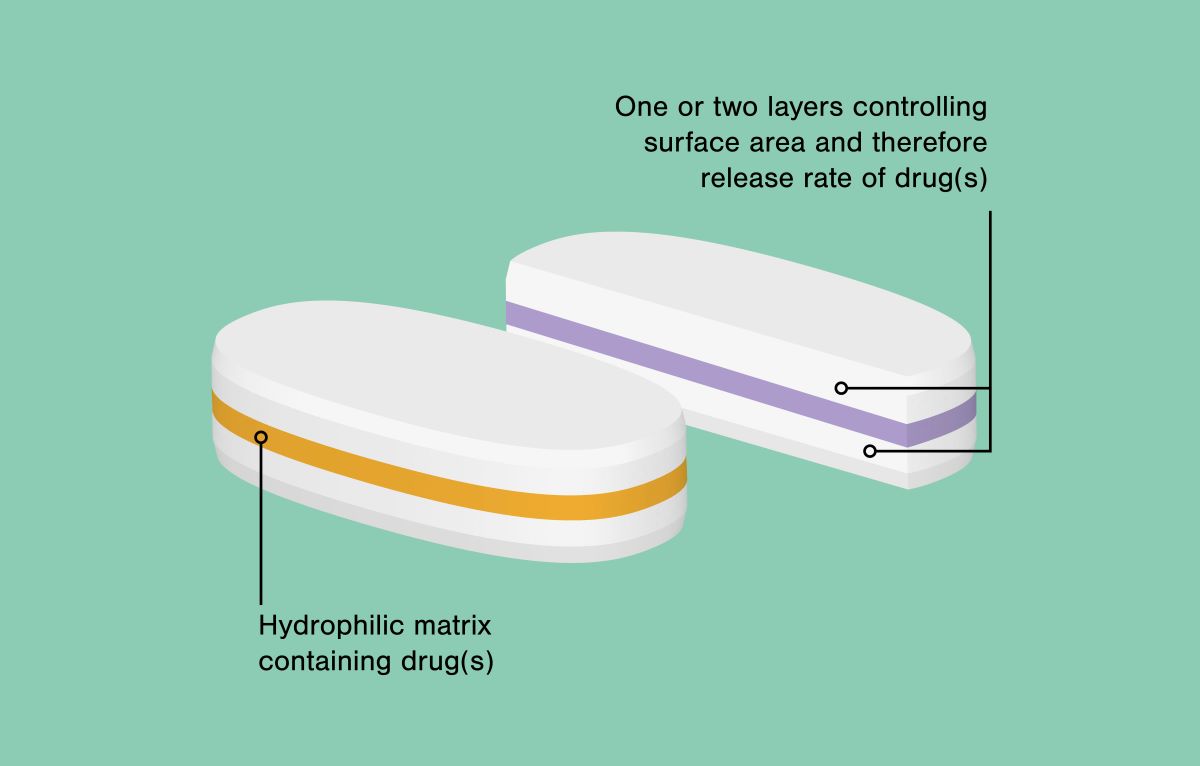
Geomatrix® is highly versatile and can be applied to a wide range of different drugs to achieve a variety of different release profiles by using a multi-layered tablet that combines use of highly swellable hydrophilic polymers with dynamic control of the surface of the layer (one or more) containing the drug (one or more) that is (are) exposed to surrounding fluids.
The combination of layers, each with different rates of swelling, gelling and erosion, controls the drug release rate within the body. When swallowed, the Geomatrix® system limits the amount of drug released by controlling the surface of the drug-containing layer exposed to fluids. Other benefits include suitability for a wide range of modified release profiles, for poorly and highly soluble drugs alike, and bi-phasic release of drugs (either rapid then slow or ascending profiles) and ability to release two or more drugs at different rates.
Skyepharma can design Geomatrix® as bi-layer or tri-layer tablets, varying tablet geometry and individual layer composition and thickness to achieve a perfect match with the Target Product Profile (TPP) in, for example, maintaining and controlling release of the active ingredient over the whole upper GIT or small intestine.
Manufacturing is carried out on specialized HATA presses, equipped with online metal check and de-duster systems to ensure compliance with the highest quality standards, with three presses running in conjunction to form an overall Geomatrix® production complex with overall capacity of more than 270,000 units per hour.
Beyond specialized equipment, manufacturing multilayer tablets also required specific expertise, such as the knowhow on how to control sticking between the different layers, as well as the parameters related to the powders themselves to manage physicochemical interactions between three different mixtures, as well as machine control and piloting.
Geoclock® press-coated tablets
Geoclock® is a validated oral drug delivery technology that is ideal for delayed release formulations, allowing timed release of active ingredient from the tablet (oral chronotherapy), independent of food consumption or pH, with API release into the GIT variable to precise timescale ranging from two to 10 hours. This capability makes it ideal for applications requiring drug release during the night or when a combination of immediate release and delayed release profiles – like a pulsatile release – is required.
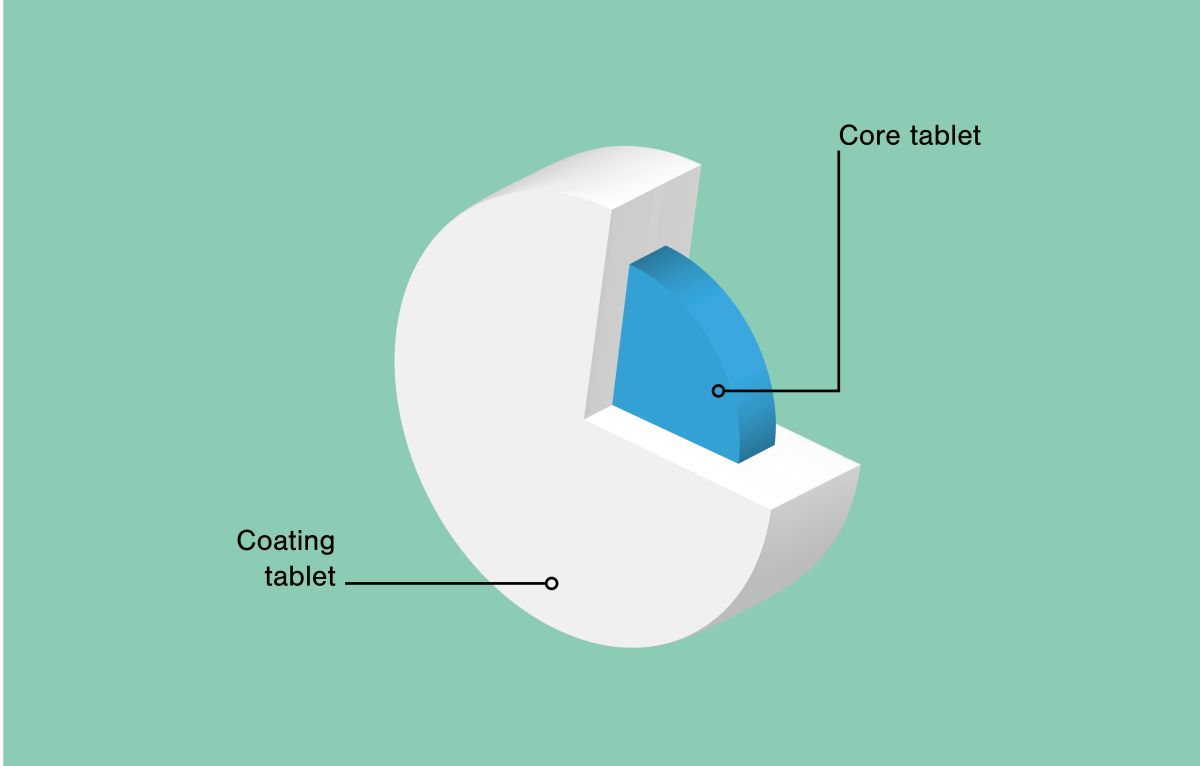
Geoclock® is a tablet in a tablet (tab-in-tab), also known as a press coated tablet or dual core tablets. APIs can be loaded into the core tablet and in the outer layer depending on the targeted release profile.
Geoclock® can additionally be used for multiple pulse delivery of one or more drugs with pre-determined time intervals between the pulses, or to target colonic release of drugs. Where more than one drug is included, the delivery profile can be different for each API.
Geoclock® is also well proven over more than a decade (both in the US and Europe) and is easily manufactured using conventional production equipment, minimizing the need for capital investment.
Formulation expertise
Skyepharma can design standard immediate release profile drugs or formulations. Skyepharma’s expert teams can also provide delayed release profiles and sustained release profiles that are both part of the broad and very diversified family of modified release formulations. These particular drug profiles can either be proposed as individual release systems or as a combination of two or more release profiles for one or more APIs – to achieve for instance the so called multipulse or chronotherapeutic drug products.
Skyepharma experts can also design drug products with a combination of immediate release and delayed release or an immediate plus a sustained release, also known as the quick-slow release pattern.
The very precise fine tuning of the release profiles is key today to answer the specific TPPs desired by the pharmaceutical companies. To successfully develop and manufacture such high complexity drug products, Skyepharma teams use the company’s proprietary technologies, backed up by very strong and well-established in-house expertise in the field of complex tablets compression, and specifically, in multilayers tablets compression and press-coated (or tab-in-tab or dual core) tablets compression.
Press-coating capabilities
When the production of the cores is based on a standard single-layer tablet compression equipment (like Skyepharma’s Fette 2100 press), a more complex machine is required for the manufacturing process of the final tablet, with Skyepharma using the Kilian S250 ZR press coater that is able to distribute and center the cores into the outer tablet, using a high-resolution sensor and high throughput capture camera to secure the detection and rejection of the off-centered tablets during the press coating process to tolerances of one tenth of a millimeter. This camera technology also allows the press coater to check core color and presence, overall tablet integrity check as well as core centering. Skyepharma’s press coater is also fitted with online metal check and de-duster systems to ensure these complex tablets production at top level quality standards.
One of Skyepharma’s great strengths is its agility and flexibility. Skyepharma expert teams are able to switch from lab scale to pilot scale and to semi-industrial and industrial size equipment according to client and project needs.
This flexibility is aided by use of Medelpharm STYL’One compression simulator that can precisely model compression profiles for each individual press including Hata multilayer presses or Kilian press coater. This allows Skyepharma development teams to troubleshoot potential production issues in advance, allowing faster scale-up and therefore securing overall drug development timelines and cost.
Resources
Click on Skyepharma as Complex Compression CDMO to learn more.