By MEGGLE Excipients
MEGGLE’s RetaLac® tablets show similar release system performance irrespective of the preparation method used
Overview
In hydrophilic sustained release systems water uptake can be primordial for the solution of an active pharmaceutical ingredient (API), as well as for the swelling of the tablet matrix impacting greatly its diffusion.
Recent studies have shown MEGGLE’s RetaLac®- co-processed Lactose and Hypromellose, especially designed for direct compression (DC) and dry granulation (DG) – to have analog water uptake kinetics and changes in tablet dimensions irrespective of the preparation method used. This similar release system performance may thus smartly help optimizing production unit operations without impacting your release profile.
Overcoming the powder technological and production-related deficiencies of frequently used single components by co-processing without compromising the complex API-matrix interaction during water imbibition was the challenge to be solved by RetaLac®.
RetaLac® is beneficial across a broad range of applications including efficient DC of sustained release formulations, DG, or e.g. convenient and rapid preparation of HPMC suspensions.
Study methods
Water uptake kinetics of tablets were determined gravimetrically using similar conditions as for in vitro drug release calculations. The tablets were weighed before exposure to the release media at time t=0. At predetermined time points samples were withdrawn, excess water carefully removed using Kimtech precision wipes (Kimberly-Clark, Kent, UK), accurately determined [wet mass (t)] and dried in an oven [Binder, Tuttlingen, Germany) at 45oC to constant mass [dry mass (t)]. The tablet water content [%] at time t was calculated as follows:
Water content [%] (t) = wet mass (t) – dry mass (t)/wet mass (t) x100%
Dynamic changes in tablet dimensions (swelling) were monitored with a micrometer gauge (Digimatic Micrometer, Mitutoyo, Tokyo, Japan). At predetermined time points samples were withdrawn and their diameter was measured in the wet state.
Under given conditions tablets with drug loadings of 20% Propranolol HCl or more could not be prepared via wet granulation due to granules with extreme hardness and variability in weight approaching the limits of the tableting machine. Finally, all experiments were conducted in triplicate.
Matrix tablets were also exposed to colored aqueous solution to allow their swelling and water uptake characteristics to be captured on video (see Resources).
Results Water Uptake Kinetics
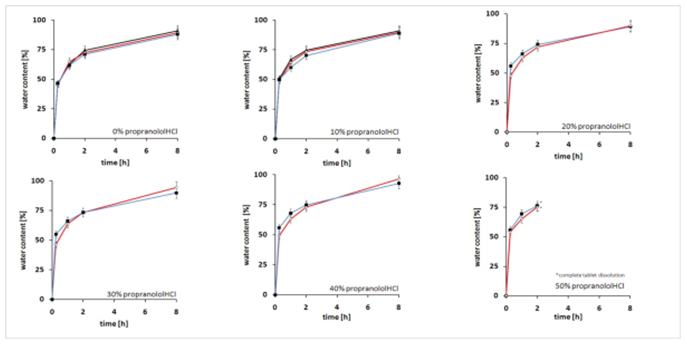
Effects of the type of preparation technique between 0 and 8 hours in release medium 0.1M HCl monitored by the water content of RetaLac®-based tablets (red line) in comparison to other tablet preparation methods as the physical blend (blue line) or the wet granulated form (black line) upon different concentrations of Propranolol HCl between 0 and 50% drug load. Little difference was observed, indicating similar release system performance. This applied also to phosphate buffer, pH 7.4 (not shown). Initial tablet heights were 2.5mm with a diameter of 11.3mm.
Results Dynamic Changes in tablet dimension (diameter)
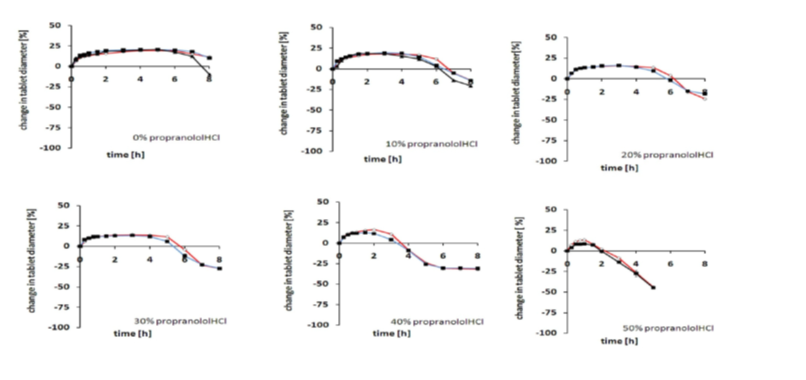
Effects of the type of preparation technique under equal testing regime investigated by dynamic changes in tablet diameter of RetaLac®-based tablets (red line) in comparison to preparation method physical blend (blue line) and the wet granulated form (black line) upon different concentrations of Propranolol HCl between 0 and 50% drug load. Little difference was observed, indicating similar release system performance. This applied also to phosphate buffer, pH 7.4 (not shown). Initial tablet heights were 2.5mm with a diameter of 11.3mm.
Resources
Click on Technical information to see detailed RetaLac® specifications.
Click on Diffusion of a RetaLac® Tablet for video


















