By PHC Europe…
PHCbi Cleanroom classification
As a pioneering and world-respected manufacturer of medical and research equipment, PHCbi understands the importance of optimizing equipment to comply with the specialized requirements of the cleanroom environments that are becoming increasingly important to the pharmaceutical and life sciences industries.
To ensure that its equipment can be trusted as an essential tool for cleanroom applications, PHCbi has invested in the certification of its flagship cold storage and cell culture incubators to meets GMP standards by being ISO cleanroom classified by independent approved testing laboratories.
Increasing Need for Cleanroom Classification
Recent successes in cell and immune-mediated therapies have accelerated the possibilities for cell-based therapies for mainstream treatments worldwide [1]. As stem cell research and regenerative medicine continue to gain clinical importance, the need for reproducibility, reliability and contamination-free performance of basic laboratory equipment is essential to the quality process.
Laboratories and other settings that work with biologically relevant products require a clean and controlled environment to establish and maintain sample integrity. Contamination events cost both time and resources, leading to significant delays in research, as well as patients’ ability to receive therapeutic treatments [2]. One of the most impactful precautions laboratories and clinical manufacturers can take in avoiding contamination is establishing and maintaining a cleanroom that meets Good Manufacturing Practice (GMP) guidelines, which apply to all phases of cell collection, processing and storage [3}.
Threats to Cell Culture-Based Product Production
Sterile or aseptic assurance is an important factor for manufacturing living cell/tissue products that cannot be sterilized. Initial cell and biological materials have inherent susceptibility to contamination from microorganisms and viral contaminants [4]. Since atmospheric air is laden with potentially infectious microparticles, maintaining proper cleanroom environment with appropriate equipment minimizes the risk of potential stressors and exposure to contaminants [5]. Cell culture contaminants can originate from other mammalian cells, microbial sources or can be from a non-living source, with any contamination of cell cultures potentially compromising results, producing genetic instability, transformation, changes in normal physiological function and changes in viral susceptibility [6], as well as presenting risk of cross-contamination to other products, creating even greater loss of resources and time.
Importance of ISO Cleanroom Classification
A cleanroom is a controlled environment where the concentration of airborne particles like dust, microbes and aerosol particles are controlled. Cleanrooms are maintained and utilized in a manner that minimizes the introduction, generation and retention of particles in the environment. All cleanrooms are classified according to the cleanliness level of the air inside them [7].
Laboratories and clinical facilities need cleanrooms for a variety of reasons. Contaminants or particles in the air greatly impact the process of both testing and manufacturing samples and products. The creation of particles by certain laboratory equipment can lead to accelerated degradation, contamination and total loss of biologically relevant material [8].
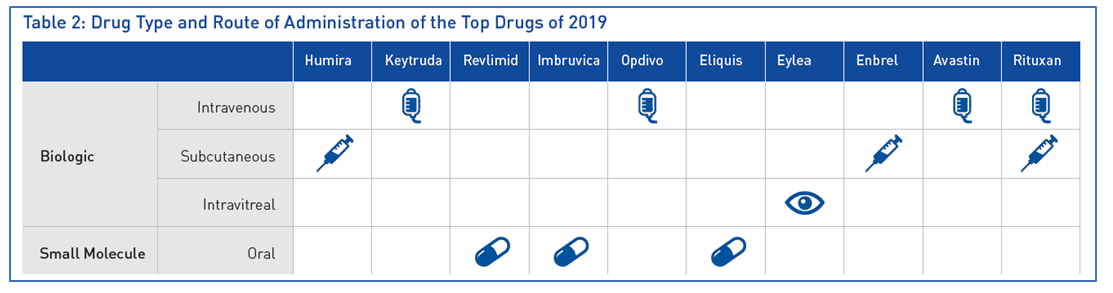
Table 2 confirms that of the top 10 best-selling drugs of 2019, seven were biologics. [9]. When manufacturing biological products, it is imperative to acknowledge that any change during the manufacturing process, regardless of how minor, may alter product quality and efficacy [10]. Therefore, within cGMP facilities, it is important to classify clean areas accurately. One of the first steps in this process is implementing cleanroom certified furniture and equipment.
ISO Cleanroom Classification
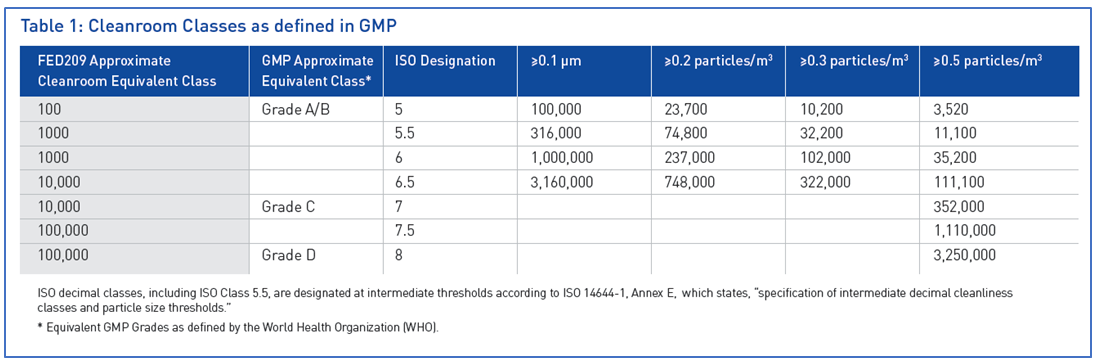
Meeting requirements for cleanrooms is one of the most critical issues for laboratories adhering to Good Manufacturing Practice (GMP) requirements, which stipulate that cleanroom classifications should be carried out according to ISO 14644-1. This ISO classification impacts every cleanroom user in the GMP community.
The International Organization for Standardization (ISO), a non-governmental body charged with promoting worldwide standards to ensure safe, reliable and high-quality products, has developed classifications associated with the levels of cleanroom certification. ISO awards cleanroom designations are based on a threshold of allowable particles within a specified area (Table 1).
ISO 14644-1 includes Part 14: ‘Assessment of suitability for use of equipment by airborne particle concentration’, developed to assess the suitability of equipment for use in cleanrooms. This section outlines the methods used to determine the total particle emission of equipment and provides data that may be used to determine the particle load on a cleanroom. For all equipment certified by an internationally recognized safety certification company, testing must adhere to the methodology of particle emission defined by Part 14. In addition, before any testing can occur, the certification company must ensure all equipment is compliant with the principles that stipulate equipment is manufactured with appropriate materials and surface finishes, avoidance of static air zones, cleanability and considerations for maintenance.
PHCbi Design for Cleanrooms
PHCbi understands that equipment not qualified for cleanroom settings can present a variety of hurdles in proper cleanroom maintenance. For example, uncertified tools and equipment with motors and other moving parts can introduce a reservoir of particles into the environment. Some essential equipment also incorporate motors and other devices that have a multitude of potential particles to shed.
PHCbi brand products have been carefully designed, engineered and constructed to fit seamlessly into any space or protocol. Engineered with cell health in mind, PHCbi brand MCO incubators have minimal moving parts and optimize decontamination protocols while minimizing contamination risks. Its ultra-low temperature (ULT) freezers are built with a unique sealed refrigeration system, coupled with a sealed bearing design on the cooling fans to reduce particle emissions.
Testing Methodology
Each unit receives appropriate ISO CleanRoom classification after undergoing the following testing methodology:
- Testing is performed in a dynamic environmental chamber with defined air, temperature, and humidity conditions.
- Cleanliness of the chamber is calculated according to ISO 14644-14:2016.
- Device is loaded in the chamber for 24 hours to find equilibrium before testing.
- Particle count concentrations are then monitored for 120 minutes, with number of particles per cubic meter measured in size ranges of ≥ 0.1 μm, ≥ 0.2 μm, ≥ 0.3 μm and ≥ 0.5 μm in diameter.
- Mean particle concentrations, upper confidence limit in the empty chamber and corresponding ISO Class ratings are then calculated and defined according to ISO 14644-1:2015.
References:
- Balendu Shekhar Jha, Mitra Farnoodian, and Kapil Bharti, “Regulatory considerations for developing a phase Iinvestigational new drug application for autologous induced pluripotent stem cells-based therapy product.” Stem Cells Translational Medicine. (August 2020): 10:198–208, https://doi.org/10.1002/ sctm. 20-0242 PMID: 32946199
- Barone, P.W., Wiebe, M.E., Leung, J.C. et al. “Viral contamination in biologic manufacture and implications for emerging therapies.” Nature Biotechnology 38, (April 2020): 563–572, https://doi.org/10.1038/s41587-020-0507-2 PMID: 32341561
- Ottria G, Dallera M, Aresu O, Manniello MA, Parodi B, Spagnolo AM, and Cristina ML. “Environmental monitoring programme in the cell therapy facility of a research centre: preliminary investigation.” Journal of Preventive Medicine and Hygiene 51, no. 4 (Dec 2010):133-8. PMID: 21553557
- Geraghty RJ, Capes-Davis A, Davis JM, et al. “Guidelines for the use of cell lines in biomedical research.” British Journal of Cancer. 111, no. 6 (August 2014): 1021-46, doi:10.1038/bjc.2014.166 PMID: 25117809
- Segeritz, Charis-P., and Ludovic Vallier. “Cell Culture: Growing Cells as Model Systems In Vitro.” Basic Science Methods for Clinical Researchers (April 2017): 151–172, doi:10.1016/B978-0-12-803077-6.00009-6
- David Pamies, Thomas Hartung. “21st Century Cell Culture for 21st Century Toxicology.” Chemical Research in Toxicology 30, no. 1 (January 2017): 43–52, doi:10.1021/acs.chemrestox.6b00269 PMID: 28092941
- “Current Good Manufacturing Practice (CGMP) Regulations.” U.S. Food & Drug Administration (FDA), last modified 09/21/2020. www.fda.gov/drugs/ pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations
- Fernando Cobo, David Grela, and Ángel Concha. “Airborne particle monitoring in clean room environments for stem cell cultures.” Biotechnology Journal, 3, no. 1 (January 2008): 43-52. doi.org/10.1002/biot.200700122 PMID: 18034434
- Yip, Stephanie. 2021. Pharmaintelligence.Informa.Com. https://pharmaintelligence.informa.com/~/media/informa-shop-window/ pharma/2020/files/reports/top-10-best-selling-drugs-of-2019.pdf
- Moutsatsou, P., Ochs, J., Schmitt, R.H. et al. “Automation in cell and gene therapy manufacturing: from past to future.” Biotechnology Letters, 41 (September 2019): 1245–1253 doi.org/10.1007/s10529-019-02732-z PMID: 31541330
Resources
Click on Cleanrooms, GMP, and ISO Certification for further information.
Click on ISO and GMP Cleanroom Standards for further information.