By Charles Ischi…
Oral solid dosage in-process control – automate your tablet and capsule testing procedures!
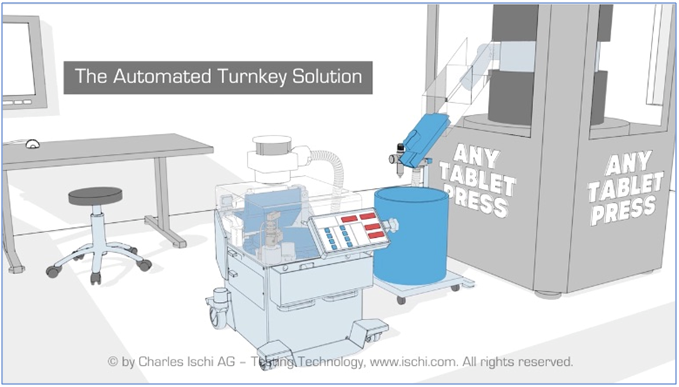
Charles Ischi AG Testing Technology offers its In-Process Control (IPC) solution as a turnkey add-on equipment that can upgrade in-process control tablet testing procedures in-line to all makes and model of tablet press and capsule filler machinery.
The solution is compact, highly mobile and will fit to any tablet press and connect to a central networked server for centralized data recording and IPC processing.
Tablet production challenges
An average tablet press has an output of 500’000 tablets/hour. During the production start up, testing is performed to set up the tablet press. Once the correct results are achieved, samples are often taken only every 30 minutes and the testing performed in the IPC lab manually. Often after initial start up of tablet production, physical property violations can occur, therefore we need to assure the uniformity of the final blend by carrying out frequent IPC checks. However, if your tablet press starts to run out of specification and testing is performed only every 30 minutes, the reject could reach in excess of 250’000 tablets or a scrapped batch!
Aside of the loss of the rejected tablets (the end-price of API’s and finished tablets can be enormous), you also have to calculate other costs like scrap, waste, re-work of material. It can take you hours or even days! Downtime equals lost revenue!
Now imagine the benefits of taking tablet samples for testing in shorter intervals, for example every 5, 10 or 15 minutes. Automatically, once a trend in weight, thickness or hardness gain/loss is detected at an early stage, the operator has the possibility to check the tablet press and adjust. If the tablet tester detects out of specification results, it sends a stop signal to the tablet press.
Tablet production using conventional presses suffer from a number of common drawbacks and challenges. These include:
- Need for time and labour intensive manual testing
- Operator time
- High reject rates due to long reaction times
- Data recording errors
- Generation of copious paper documentation
- Risk of cross contamination between batches
All of these factors add to the amount of time, labour and cost required for tablet production, as well as risks in process errors.
Charles Ischi IPC solution features
The Charles Ischi IPC tablet testing solution is a compact and mobile modular system that can ‘plug and play’ fit to any current tablet press. Features include:
- Sampling gate for single and double sided tablet presses
- Air transport system for clean and efficient sampling
- Multi-parameter tablet analysis
- Data acquisition and analysis software
- 21 CFR part 11 cGMP – data integrity
- High frequency testing triggered by time or quantity
Automatic tablet testing capabilities and benefits
Automated & frequent quality control enables the system to recognize trends in weight, hardness, thickness, diameter/length and width variation in tablets.
The system generates audio or visual alerts and automatic stops when specified tolerance limits are reached, allowing operators to adjust the tablet press to prevent rejects in production.
Connective and compliant
Time or volume triggered tablet sampling allows frequent quality control of the physical characteristics of tablets.
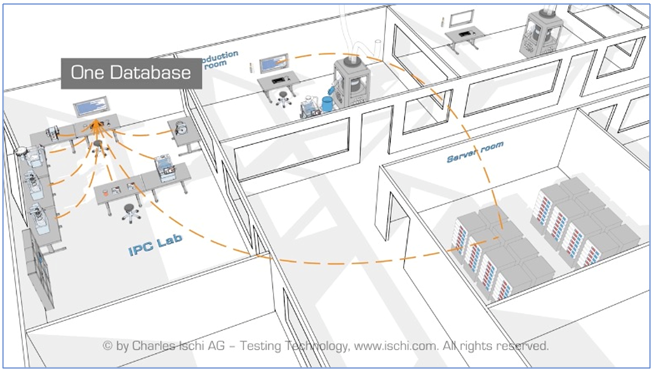
The system sends testing results to a central server, generating IPC data that can be applied to full production.
This allows the operator to manage product recipes and collect test data from all physical testing devices in one shared database for comprehensive batch records.
The system is compatible with OPC, LIMS and MES protocols for full connectivity while the Software ensures full 21CFR part11 GMP compliance, also allowing full archiving of all results and product recipes, with electronic batch reports.
IPC tablet testing benefits
The Charles Ischi IPC tablet testing solution delivers a series of tangible benefits that overcome all the functional challenges of tablet production:
- Speed up tablet development
- Reduce risk and reject rates
- Save valuable operator time
- Reduce human errors
- Avoid cross contamination
- Rationalize and reduce batch record documentation
- Ensure full 21 CFR part 11 compliance
The system is shown in a video that demonstrates its main features and functions (see Resources).
Resources
Click on Automatic Tablet Testing to view video.