By Bachem AG
Opioid peptides
Bachem offers a wide selection of opioid peptides and opioid research-related products that support advances in studies of addictive and emotional behaviors, the regulation of pain and stress responses and control of food intake.
Endogenous opioid peptides and their receptors from promising therapeutic targets for the management of pain but also offer starting points for the treatment of emotional disorders and addictive diseases.
Background
Opioid receptors are expressed in various tissues and cell types. They are found in central and peripheral neurons, but also in other cell types such as neuroendocrine, immune, and mucosal cells.
The discovery of the classical opioid receptors and the first endogenous opioid peptides, Met- and Leu-enkephalin, dates back to the early 1970s. Subsequent work has led to the identification of further opioid peptides and the cloning of their receptors.
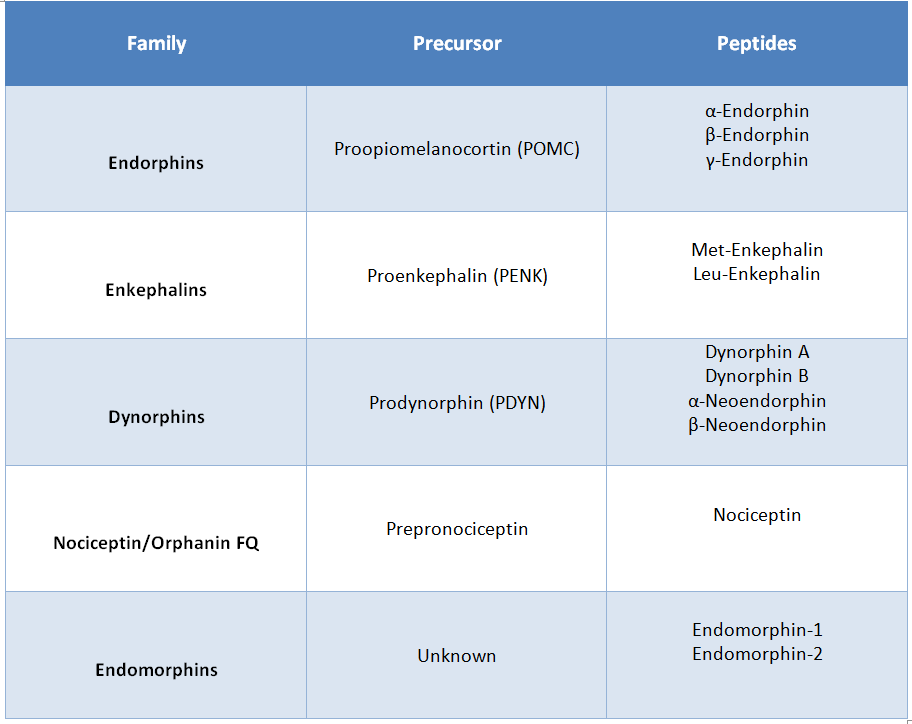
The endogenous opioid peptides are relatively short peptides and include the endorphins, enkephalins, dynorphins, and nociceptin/orphanin FQ. They are mainly produced in the central nervous system (CNS) but also in various glands such as the pituitary gland and the adrenal glands.
The endorphins are cleaved from proopiomelanocortin (POMC) which is also the precursor protein for other peptides such as ACTH, β-lipotropin, α-MSH, β-MSH, and γ-MSH. Met-enkephalin and Leu-enkephalin are produced from the precursor protein proenkephalin A that gives rise to additional peptides including two C-terminal extended forms of Met-enkephalin. Dynorphin A and B as well as α- and β-neoendorphin are derived from prodynorphin (PDYN) also known as proenkephalin B. The heptadecapeptide nociceptin/orphanin FQ is derived from prepronociceptin.
Opioid receptors
The opioid receptors belong to the superfamily of the seven-transmembrane G protein-coupled receptors (GPCR). They consist of the classical μ-, δ-, and κ-opioid receptors (MOP, DOP, KOP) and a fourth member, the nociceptin/orphanin FQ receptor (NOP). NOP receptors share high structural homology with the other opioid receptors but do not bind opioids. On the other hand, its ligand, nociceptin/orphanin FQ, shows no affinity for the classical μ-, δ-, and κ-opioid receptors.
Nano opioid (μ-opioid) receptors represent the primary molecular target for morphine and mediate both the beneficial (analgesia) and adverse effects such as respiratory depression of this drug. They also mediate the rewarding properties of non-opioid drugs such as alcohol, cannabinoids and nicotine. Stimulation of κ-opioid receptors results in dysphoria and other aversive effects such as hallucination. Activation of δ-opioid receptors regulates emotional responses and causes anxiolytic and antidepressant effects. NOP activation has a modulatory role on μ-opioid receptor-mediated actions. NOP receptors are additionally involved in motor control and other central processes.
In contrast to the other opioid peptides, no precursor peptide or gene has been identified for endomorphin-1 and-2 (See Table 1).
Opioid Receptor Signaling
The endogenous opioid peptides exhibit different affinity and selectivity for the various opioid receptor subtypes (See Table 2).
All four opioid receptors couple to the inhibitory G proteins (Gαi and Gαo). They convey extracellular signals within the cells by activating the heterotrimeric G proteins, which interact with the cytoplasmic domains of the receptors. Upon activation, the Gα and Gβγ subunits dissociate from one another, allowing them to act upon their downstream effectors and thereby initiate a variety of intracellular signaling responses. These include suppression of adenylate cyclase activity by Gα subunits, thereby reducing cyclic AMP production, and inhibition of voltage-gated calcium channels (VGCC) as well as activation of G protein-gated inwardly rectifying potassium (GIRK) channels by Gβγ subunits.
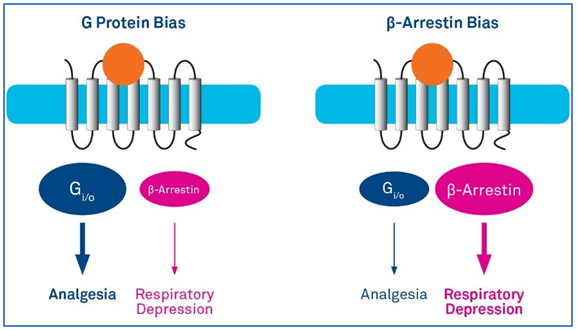
Receptor activation also results in receptor phosphorylation by G protein-coupled receptor kinases, which increases its affinity for β-arrestin molecules. β-Arrestin molecules are involved in the regulation of G protein signaling through receptor desensitization and internalization. They also act as scaffolds, bringing together other proteins to activate separate signaling pathways including the mitogen-activated protein kinase (MAPK) cascade. Biased opioid receptor ligands stabilize receptor conformations preferentially activating either the G protein or the β-arrestin pathway (Figure 1). The discovery of this type of ligands has opened a new area of drug research, aiming for example, at the design of drugs with less side effects.
There is also evidence that opioid receptors can form heterodimers composed of different opioid receptors, which offers the potential to develop bivalent ligands with unique pharmacological properties.
Exorphins
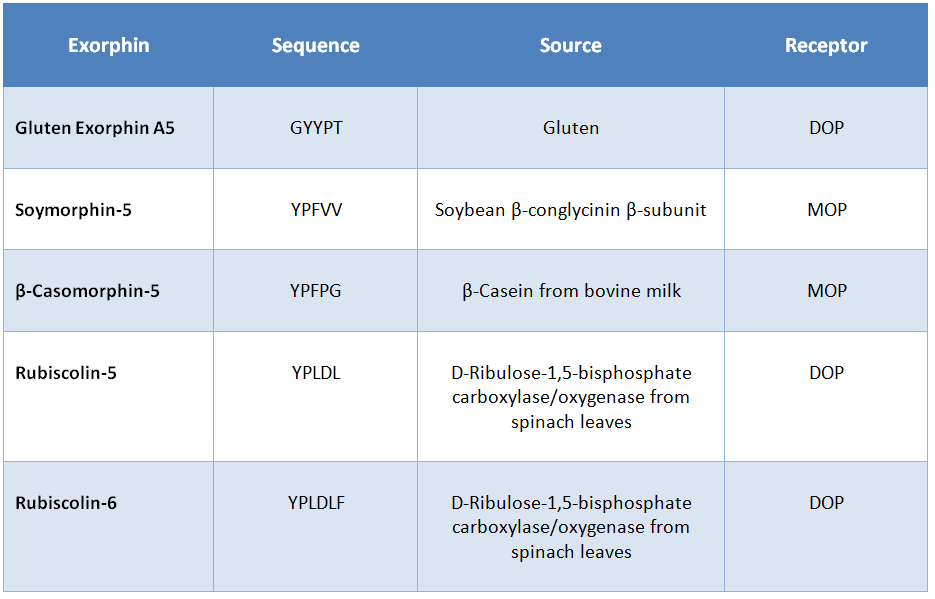
Exorphins, also known as exogenous opioid peptides, are short food-derived peptides. They can arise during digestion through the enzymatic cleavage of proteins by enzymes such as pepsin, trypsin and chymotrypsin. The most common food sources for these bioactive peptides are milk or dairy products, soybean, cereals, and green vegetables such as spinach. (See Table 3).
The physiological significance of these peptides remains controversial. They have been implicated in autism and schizophrenia and might be involved in emotional behavior and memory processes by influencing hormonal or neurotransmitter systems via opioid receptors.
Opioid Peptides in Clinical Development
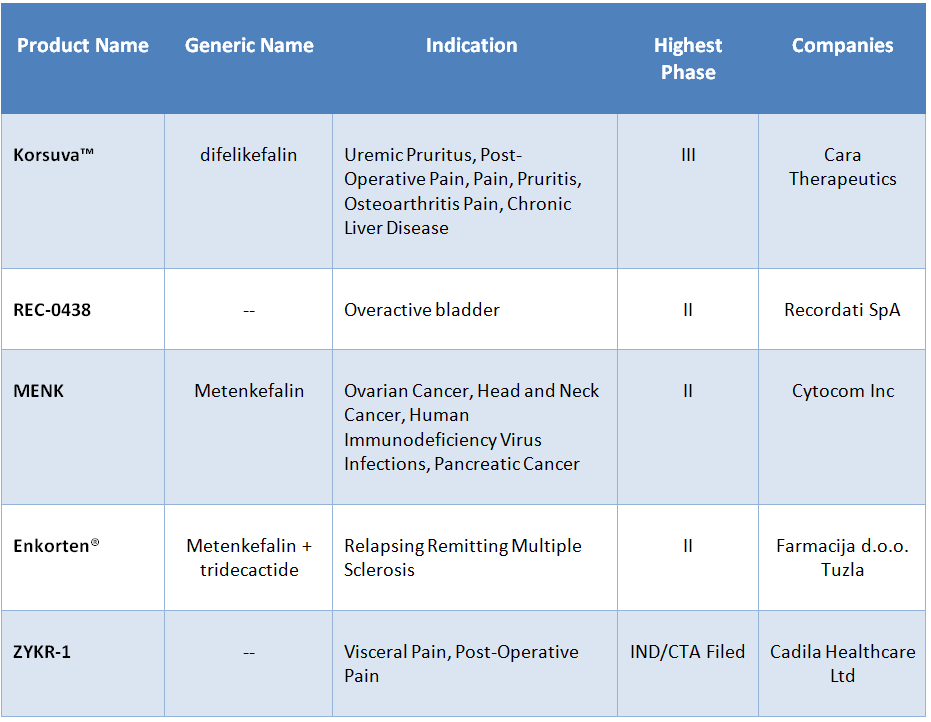
Opioid peptides are known to play a role in analgesia, feeding, memory, motivation, immune responses and a variety of other functions. Opioid receptors serve as important drug targets and opioid peptide analogs are of interest for the treatment of pain and other conditions such as mood disorders and substance abuse (17). For example, peptide-based analgesics are being explored in the search for safer alternatives to existing opioid drugs because they may produce less tolerance, respiratory suppression, and addiction. There are several promising opioid peptides in clinical development for various indications as shown in Table 4.
-
- Phase III Candidates:
- Cara Therapeutics is developing Korsuva™, a κ-opioid receptor agonist, for the treatment of post-operative pain, uremic pruritus, chronic and acute pain, and chronic liver disease. In January 2019, Cara Therapeutics completed enrollment of the KALM-1 Phase III trial of Korsuva in hemodialysis patients with pruritus (18).
- Phase III Candidates:
-
- Phase II Candidates:
- REC-0438 is under development at Recordati SpA for the treatment of overactive bladder in pediatric spina bifida patients with spinal lesions and neurogenic detrusor overactivity. This product candidate is a nociceptin analog that acts by agonizing the Opioid Receptor-Like 1 (OPRL1) receptor. In 2018, Recordati registered a Phase II trial to study the effects of two different doses of REC-0438 in patients with neurogenic detrusor overactivity (NDO) due to spinal cord injury (18).
- Cytocom is developing MENK, a potent opioid peptide, which influences DNA synthesis and cell growth. Research studies have shown that MENK can inhibit cancerous cell growth and boost the immune system (19). The candidate is in Phase II development for pancreatic cancer, ovarian cancer, hepatocellular carcinoma, head and neck cancer, and Human Immunodeficiency Virus (HIV) Infections (AIDS). MENK treatment is administered by first isolating a patient’s lymphocytes and incubating them with IRT-101. Following incubation, the lymphocytes are re-infused back into the patient to kill tumor cells (18).
- Enkorten®, a combination of metenkefalin and tridecactide, is under development at Farmacija d.o.o. Tuzla for the treatment of relapsing remitting multiple sclerosis (RRMS). Metenkefalin targets the δ-opioid receptor and tridecactide targets the melanocortin receptor. In 2017, a Phase IIIb trial was registered which will evaluate the efficacy, safety and tolerability of Enkorten in patients with RRMS (18).
- Phase II Candidates:
- Phase I Candidates:
- Cadila Healthcare is developing ZYKR-1 for the treatment of visceral pain and post-operative pain. ZYKR-1 acts as a κ-opioid receptor agonist. In 2018, the company registered a Phase I study of ZYKR-1 to evaluate the safety, pharmacokinetics and pharmacodynamics in healthy volunteers. The trial will take place in India (18).
Research and development surrounding opioid peptides has expanded from emphasis on pain regulation to include other indications such as cancer and RRMS. For researchers and organizations working on the research and development of opioid peptides, Bachem offers a wide selection of opioid peptides, a comprehensive custom peptide synthesis service, and the production of New Chemical Entities.
References
- G.Corder, D.C.Castro, M.R.Bruchas, G.Scherrer, Endogenous and exogenous opioids in pain. Annual Review of Neuroscience 2018, 41, 453-473.
- A.Stefanucci, A.Mollica, G.Macedonio, G.Zengin, A.A.Ahmed, E.Novellino, Exogenous opioid peptides derived from food proteins and their possible uses as dietary supplements: A critical review. Food Reviews International 2018, 34, 70-86.
- R.J.Valentino, N.D.Volkow, Untangling the complexity of opioid receptor function. Neuropsychopharmacology 2018, 43, 2514-2520.
- J.M.Wenzel, J.F.Cheer, Endocannabinoid regulation of reward and reinforcement through interaction with dopamine and endogenous opioid signaling. Neuropsychopharmacology 2018, 43, 103-115.
- R.I.Anderson, H.C.Becker, Role of the dynorphin/kappa opioid receptor system in the motivational effects of ethanol. Alcoholism, Clinical and Experimental Research 2017, 41, 1402-1418.
- K.M.Olson, W.Lei, A.Keresztes, J LaVigne, J.M Streicher, Novel molecular strategies and targets for opioid drug discovery for the treatment of chronic pain. Yale Journal of Biology and Medicine 2017, 90, 97-110.
- S.Spangler, M.R.Bruchas, Tuning biased GPCR signaling for physiological gain. Cell 2017, 171, 989-991.
- J.S.Smith, S.Rajagopal, The beta-arrestins: Multifunctional regulators of G protein-coupled receptors. The Journal of Biological Chemistry 2016, 291, 8969-8977.
- L.Toll, M.R.Bruchas, G.Calo, B.M.Cox, N.T.Zaveri, Nociceptin/Orphanin FQ Receptor Structure, Signaling, Ligands, Functions, and Interactions with Opioid Systems. Pharmacological reviews 2016, 68, 419-457.
- J.McDonald, D.Lambert, Opioid receptors. BJA Education 2014, 15, 219-224.
- E.E.Benarroch, Endogenous opioid systems. Neurology 2012, 79, 807.
- D.S.Sauriyal, A.S.Jaggi, N.Singh, Extending pharmacological spectrum of opioids beyond analgesia: multifunctional aspects in different pathophysiological states. Neuropeptides 2011, 45, 175-188.
- B.A.Gosnell, A.S.Levine, Reward systems and food intake: role of opioids. International Journal of Obesity 2009, 33, S54.
- B.L.Kieffer, Opioid peptides and receptors. In: Squire LR (ed). Encyclopedia of Neuroscience. Academic Press: Oxford, 2009, 235-240.
- A.Koneru, S.Satyanarayana, S.Rizwan, Endogenous opioids: Their physiological role and receptors. Global Journal of Pharmacology 2009, 3, 149-153.
- J.C.Froehlich, Opioid peptides. Alcohol Health & Research World 1997, 21, 132-136.
- J.V.Aldrich, J.P. McLaughlin, Opioid Peptides: Potential for Drug Development. Drug Discov. Today Technol. 9, e23-e31 (2019)
- GlobalData (2019)
- MENK. Cytocom (2019)
Resources
Click on Bachem opioid peptides to browse Bachem online store.
Click on Bachems News to see latest News & Events.