By Molnár-Institute for…
MOLNÁR-INSTITUTE helps pioneer combination methodology for high throughput RPLC analysis of COVID-19 antibodies
Berlin: – The Molnár-Institute for Applied Chromatography has played a leading role in a pioneering study examining application of an innovative methodology to speed up the chromatographic characterization of anti-COVID protein therapeutics using a combination of automated, systematic modeling and ultra-short liquid chromatography (LC) columns.
The study has yielded highly encouraging results across a range of chromatographic techniques, including reverse phase liquid chromatography (RPLC) [1].
Response to COVID-19
The study, led by Bastiaan Duivelshof from the University of Geneva, also involved Molnár-Institute application specialist Arnold Zöldhegyi and Waters Corporation scientist Szabolcs Fekete. It was launched as a joint response to the need exposed by the COVID-19 pandemic to accelerate emergency use authorization (EUA) of new therapeutics and vaccines that included several monoclonal antibodies (mAbs).
Since EUA submissions usually require scientific data on drug candidates to be collated in parallel with drug development, there is little time to spare for misguided method development or waiting on traditional turnaround times for LC characterizations.
The team took this as their cue to investigate new means of expediting rapid chromatographic characterization of protein therapeutics that combined the use of automated, systematic modeling and state-of-the-art ultrashort LC columns to quickly optimize high throughput RP separations for two exemplar COVID-19-related mAbs: casirivimab-imdevimab cocktail and tocilizumab.
Combination method
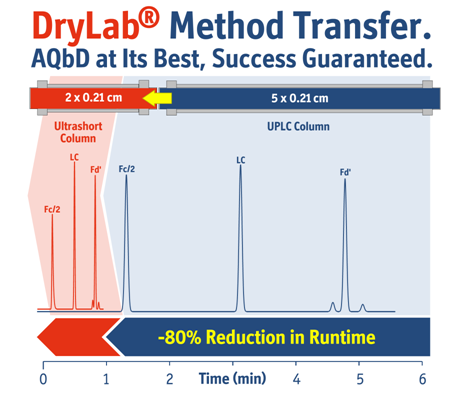
The study demonstrated that using the new combination method, both studied mAb-samples could be characterized by RPLC analysis in as little as a sub-two-minute separation run time; making it feasible to run the common suite of four LC profiling techniques (RP, IEX, HILIC and SEC) within a one-hour turnaround time (Fig. 1).
Untapping the full resolving potential of the new column hardware and speeding up the entire analytical process, computerized modeling—Molnár-Institute’s DryLab® software platform—played a crucial role by effectively reducing the number of wet-lab experiments and quickly contextualizing complex chromatographic data by acquired Design Spaces [2].
The other main innovation used in the study was a streamlined analytical quality-by-design (AQbD) modeling workflow based on automated generic approaches (Design-of-Experiments) for monoclonal antibody and antibody-drug-conjugate separations. This takes into account previous scientific results that reveal the propensity of large proteins to be extremely sensitive to the mobile phase composition, becoming eluted in many types of separations with only a very minor change in eluent strength, referred to as the “on-off” phenomenon or “bind and elute” retention mechanism. The prevalence of this effect increases with molecular size (hydrophobic surface) during RPLC. A consequence of “on-off” elution behavior is that only the first segments of a column will actively take part in peak retention and separation, which means very short (15–20 mm) columns can be used to speed up separations without compromising efficiency (Fig. 2).
RPLC results
The RPLC analysis of casirivimab and imdevimab cocktail’s subunits confirmed a pronounced trend in RPLC analysis of mAbs that favors a so-called “middle-up”- approach, which consists of a separation of reduced light chain (LC) and heavy chain (HC) variants or the analysis of Fc, sFc, Fab, F(ab’)2 or Fd’ fragments that can be created by limited proteolysis [3].
RPLC commonly used to detect various chemical and post-translational modifications via a subunit level analysis, usually when separations performed at temperatures above 60◦C to obtain sharpened peak shapes and improved recoveries.
In the study, the effect of mobile phase temperature and gradient steepness were systematically studied based on DryLab® 2D critical resolution maps. In case of all four samples (reduced casirivimab, reduced imdevimab, digested and reduced casirivimab, and digested and reduced imdevimab), mobile phase temperature had only a slight impact on resolution while gradient steepness was confirmed as the critical variable.
The study team found 75◦C as a good compromise of temperature, yielding complete peak recovery and relatively narrow peak widths. They also found that linear gradients of 26–36 %B (for the digested and reduced samples) and 29–37 %B (for the reduced samples) provided nearly uniform selectivity for the main species and acceptable resolution of some minor variants.
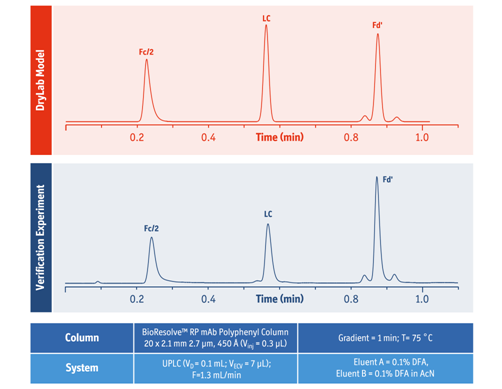
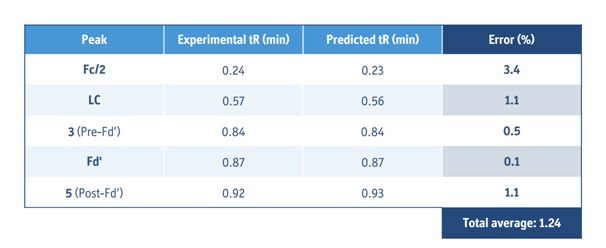
Using the separation models, these gradients were flawlessly in-silico transferred from 50´2.1 to a 20´2.1 mm column with system dwell volume of 0.110 mL. Experimental verification showed perfect agreement between the virtually transferred and measured chromatograms, with average error of retention time prediction under 1.5 %. As a final step of optimization, gradient time was further decreased along with a proportional increase of the flow rate to quicken the analysis time. In the end, it was found that a gradient time of 1 minute at 1.3 mL/min flow rate provided good separation for all subjected mAb samples (Fig. 3).
Conclusions
The team identifies practical benefits of combining retention modeling and automated experiments with use of short protein columns for mAb analysis.
“Retention modeling enables the in-silico optimization of mAb (or any protein) separations within a few minutes. The automation module of DryLab® Software significantly simplifies manual work and thus speeds up the method development procedure,” the study concludes.
It suggests that the whole process to develop and optimize four chromatographic methods (also including IEX, HILIC and SEC) for two mAb samples might require only two days of experimental work, compared with at least a week using current methodology. By using short protein columns (15–20 mm long for RPLC and IEX), analysis times in the range of 1–2 min were obtained, marking a significant time savings in comparison to 10–30 min long traditional methods.
“Overall, we have shown that conventional chromatographic techniques can quickly be transferred in silico to short column, higher throughput parameters and to low dispersion chromatographic systems without the need for further experiments. These approaches might be valuable for turning around scientific data on drug candidates during critical times when the preparation of filing information must be accelerated,” the study concludes.
References
- Duivelshof, B., Zöldhegyi, A., Guillarme, D., Lauber, M. and Fekete, S. (2022). Expediting the chromatographic analysis of COVID-19 antibody therapeutics with ultra-short columns, retention modeling and automated method development. Journal of Pharmaceutical and Biomedical Analysis, [online] 221, p.115039. doi:10.1016/j.jpba.2022.115039.
- den Uijl, M.J., Schoenmakers, P.J., Pirok, B.W.J. and Bommel, M.R. (2020). Recent applications of retention modelling in liquid chromatography. Journal of Separation Science, 44(1), pp.88–114. doi:10.1002/jssc.202000905.
- Fekete, S., Rudaz, S., Fekete, J. and Guillarme, D. (2012). Analysis of recombinant monoclonal antibodies by RPLC: Toward a generic method development approach. Journal of Pharmaceutical and Biomedical Analysis, 70, pp.158–168. doi:10.1016/j.jpba.2012.06.021.
About Molnár-Institute
Founded in 1981, Molnár-Institute develops DryLab®4, a software for UHPLC modelling for a world-wide market. Its powerful modules gradient editor, peak tracking, automation, robustness and Design Space Comparison allow for the most sophisticated method development as required across modern pharma industries. Analytical scientists use DryLab®4 to understand chromatographic interactions, to reduce analysis time, to increase robustness, and to conform to Analytical Quaity by Design (AQbD) principles, according to the recently published ICH Q14 regulatory framework.
The Molnár-Institute is a registered partner of the US-FDA, CDC and many other regulatory bodies. DryLab®4 pioneered AQbD long before regulatory agencies across the world encouraged such submissions. Widely implemented by thought leaders, the software contributes substantially to the paradigm shift towards a science and risk driven perspective on HPLC Quality Control and Assurance.
Further information at: http://www.molnar-institute.com/
Resources
Click on Expediting the chromatographic analysis of COVID-19 antibody therapeutics with ultra-short columns, retention modelling and automated method development (2022) to access the full study.
Click on Contact Molnár-Institute to request other resources.