By Bachem AG
Histone peptides
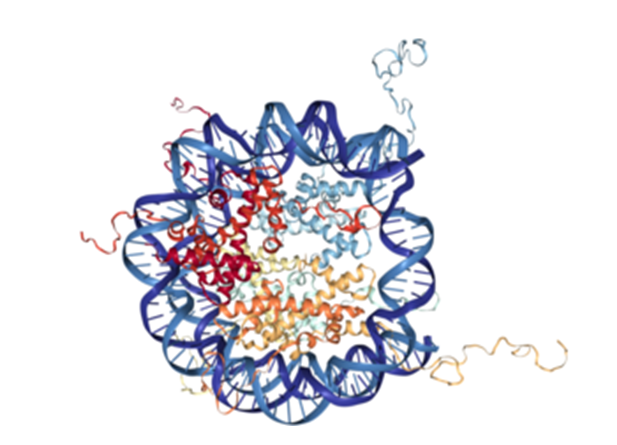
The chromosomes of eukaryotes consist of two elements; chromatin, a complex formed by DNA molecules, and proteins called histones. In order to bind efficiently to the negatively charged DNA via electrostatic interactions, histones need to be charged positively. Therefore, these peptides are especially rich in the basic amino acids lysine and arginine. In chromatin, the double helix structure of DNA wraps around a core of histones, with certain areas of the histones protruding from the formed “bead”, the nucleosome.
Acetylated histones do not only play a vital part in the transcription of DNA. Their presence or absence is also associated with human diseases, such as inflammations, addictions and cancer.
Histone families
There are varieties of different histones, which are represented by different colors within the core of the nucleosome (see illustration).
The various histones can be organized in five different families: H2A, H2B, H3, H4 and H1/H5, which in turn can be grouped into core histones (H2A, H2B, H3 and H4) and linker histones (H1 and H5). As the name suggests, core histones form the central part of the nucleosome, whereas linker histones connect the nucleosomes to form the so-called “Beads-on-a-String”-structure.
Post-translational modifications
Chromatin is not a rigid structure, but is rather flexible. If chromatin is in a loose conformation, the chromosomal DNA can be transcribed. Conversely, if chromatin is condensed (creating a complex called heterochromatin), it is inactive, and DNA transcription does not occur. An important factor governing the structure of chromatin is reversible, post-translational modification of histones. Possible modifications are:
- N-Methylation (Arg and Lys)
- N-Acetylation
- Biotinylation
- Ubiquitinylation
Formation of inactive heterochromatin is marked by methylation of a particular lysine residue (K9) on a specific histone (H3). Active chromatin on the other hand is generally associated with methylation of K4 on the same histone as well as acetylation of lysine residues in general. While the activation of chromatin via acetylation is performed by histone acetyltransferase (HAT), its deactivation is facilitated by histone deacetylases (HDACs). Study of histone acetylation is therefore closely linked to the study of these two types of enzymes.
The importance of histones
Acetylated histones do not only play a vital part in the transcription of DNA. Their presence or absence is also associated with human diseases, such as:
- Inflammatory diseases: Inhibition of HDACs leads to increased presence of acetylated histones and increased expression of inflammatory genes, after activation with inflammatory stimuli.
- COPD: In chronic obstructive pulmonary disease, a reduction of HDAC activity can be observed in the lung parenchyma, corresponding to the severity of the disease. Similarly, increase in HAT has been observed in bronchial biopsies from patients with asthma.
- Addiction: Chronic drug use, such as cocaine or alcohol abuse, initiates decreased HDAC activity in reward and learning-related brain regions (nucleus accumbens, prefrontal cortex or hippocampus). Acute administration of alcohol or cocaine on the other hand briefly increases the acetylation of H3, highlighting the mechanistic differences between acute and chronic covalent chemical changes. The transition between occasional drug use and its chronic counterpart is potentially caused by a single class of HDACs. HDAC5 is a deacetylase, which is phosphorylated and subsequently expelled from the nucleus upon stimulation by either drug use or stress. Through this decreased deacetylation, gene transcription and behavioral adaption can take place. HDAC5 is generally regenerated within 24 hours. However, during chronic stimulation this regeneration cannot take place, leading to an increased expression of stress- and addiction-associated genes, such as ΔFosB.
- Cancer: Abnormal alterations in histone acetylation are associated with the development of cancer. For instance, loss of acetylation at Lys16 of H4 is reported as a common abnormality in human cancer and low levels of acetylation at Lys18 of H3 was found to be a predictor of poor survival rates in pancreatic, breast, prostate and lung cancer. Thus, contrary to inflammatory diseases, inhibition of HDACs has been associated with a beneficial effect in the treatment of cancer. Several HDAC inhibitors have been approved by the FDA for the treatment of cancer:
-
- Vorinostat (cutaneous T-cell lymphoma)
- Belinostat (peripheral T-cell lymphoma)
- Panabiostat (multiple myeloma)
- Romidepsin (cutaneous T-cell lymphoma)
Modification with larger molecules
Contrary to the modifications with small substituents (i.e. methylation and acetylation), post-translational modifications occur much more rarely with larger molecules such as biotin and ubiquitin, which can be classified as proteins.
Nevertheless, these modifications play an important role for cellular processes. For instance, poly-ubiquitylation commonly flags proteins for degradation, while mono-ubiquitylation of H2A and H2B has been associated with regulation of gene expression.
Histone biotinylation is an especially rare post-translational modification, with less than 0.001% of human histones H3 and H4 being biotinylated in nature. While rare, this modification, too, has significant biological importance.
For instance, fibroblasts of patients suffering from holocarboxylase synthetase deficiency show severely lowered rates of histone biotinylation. There is also evidence that biotinylation is part of the cellular response to DNA damage, since the global biotinylation increases upon formation of thymine dimers in lymphoid cells (caused by UV irradiation). The role of biotinylation in signaling DNA damage is further corroborated by the rapid decrease in biotinylation of Lys12 in H4 upon double-stranded DNA breaks.
Bachem histone portfolio
Bachem offers various biotinylated histone sequences in its catalog and biotinylation is a standard modification, which can be offered as part of its custom synthesis service.
Due to the importance of histone acetylation for disease research, Bachem not only offers acetylated histone sequences but also HDAC substrates as part of its catalog portfolio.
References
P.J.Barnes, I.M.Adcock, K.Ito, Histone acetylation and deacetylation: importance in inflammatory lung diseases. Eur. Respir. J: 25, 552-563 (2005)
N.Kothapalli et al., Biological functions of biotinylated histones. J. Nutr. Biochem: 16, 446-448 (2005)
I.Cohen, E.Poreba, K.Kamieniarz, R.Schneider, Histone modifiers in cancer: friends or foes? Genes Cancer 2: 631-647 (2011)
T.Kuroishi, L.Rios-Avila, V.Pestinger, S.S.Wijeratne, J.Zempleni, Biotinylation is a natural, albeit rare, modification of human histones. Mol. Genet. Metab: 104, 537-545 (2011)
L.N.Hitchcock, K.M.Lattal, Histone-mediated epigenetics in addiction. Prog. Mol. Biol. Transl. Sci.:128, 51-87 (2014)
M.M.Muller, T.W.Muir, Histones: at the crossroads of peptide and protein chemistry. Chem. Rev.: 115, 2296-2349 (2015)
J.L.Cadet, Epigenetics of stress, addiction, and resilience: Therapeutic implications. Mol. Neurobiol: 53, 545-560 (2016)
T.Eckschlager, J.Plch, M.Stiborova, J.Hrabeta, Histone deacetylase inhibitors as anticancer drugs. Int. J. Mol. Sci: 18, (2017)
Resources
Click on Bachem histone sequences for product information.
Click on Bachem HDAC substrates for product information.
Click on Bachems News to see latest News & Events.