By Evolve Ltd.
Evolve signs ethical cannabis-focused European Distributor Partnership with small scale packaging specialist ATG Pharma
Luqa, Malta: – Life sciences environment specialist Evolve Ltd (Evolve) has finalized a partnership agreement with ATG Pharma to act as European distributor for ATG’s cGMP ethical cannabis-based products.
Building on Europe’s estimated legal medical cannabis market growth potential, this partnership provides Evolve and ATG Pharma a solid base to expand in both European and global markets.
Growing European cannabis markets
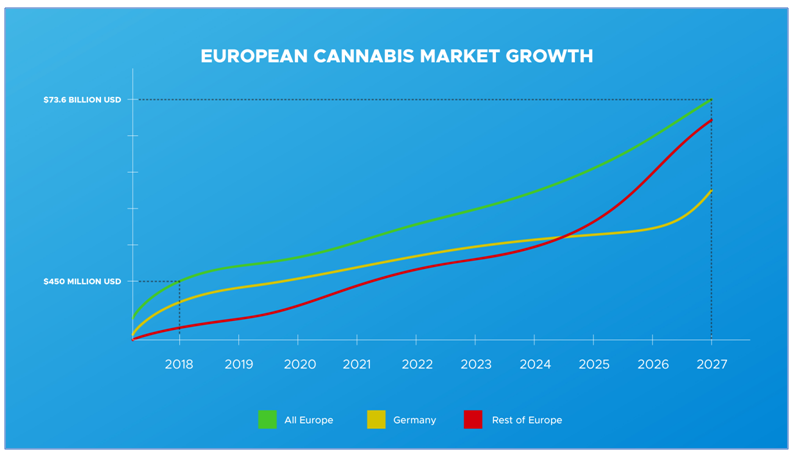
Over the next ten years, the legal cannabis industry will see tremendous market growth around the globe. Spending on legal cannabis worldwide is expected to reach $73.6 billion USD by 2027, according to a recent report by Grand View Research, Inc. (see Resources).
Currently, only a few European countries have legalized cannabis for medical purposes. However, as more European countries enact legislation to make cannabis legal for medicinal, food, cosmetic and other applications, EU sales of cannabis-derived products are projected to surpass the North American market.
Pioneering EU-GMP cannabis production
In 2018, Malta enacted legislation that allows provisions towards local production of cannabis for medical and research purposes. Following the legislative amendments, licensed medical practitioners can prescribe cannabis-based products produced under the European Good Manufacturing Practice (EU-GMP).
In 2019, Evolve helped developed the first EU-GMP-compliant medical cannabis facilities in Malta, drawing on its expertise in providing custom-made solutions for new lab supplies or specialist scientific equipment for international customers, including major pharmaceutical companies, hospitals, universities and public laboratories. Evolve included ATG Pharma bench-scale production filling and packaging platforms as part of its solution.
Milestone agreement
Chris Shar, Vice President of Sales & Business Development at ATG Pharma, commented: “This partnership is an exciting milestone for our company, as ATG is building the infrastructure to support clients on a global scale. Current revenues outside of North America account for approximately 10 per cent of total revenues, and as the Global Cannabis industry continues to expand to forecasted projections, we anticipate seeing rapid growth and market share in this sector.”
“In addition, this European partnership will allow ATG to support our North American Clients who plan to expand their companies into Europe. Partnering with Evolve Ltd. is an exciting endeavour as ATG recognizes the significant potential of European market growth for medical cannabis. This partnership aligns with ATG’s strategic goal of bringing our state-of-the-art equipment to a broader European market,” ATG added.
Ensuring regulatory compliance for Medical Cannabis
In a recent article for Malta’s Open Access Government online journal (see Resources), Evolve’s Managing Director, Christopher Busuttil Delbridge, identified consistency and quality control (QC) as two key challenges for ethical cannabis producers in navigating the EU’s complex regulatory approval process.
Evolve has begun producing a series of guides to cultivating, testing and licensing cannabinoids successfully for pharmaceutical, nutraceutical and other applications.
“For Cannabis to be labelled ‘Medical Grade’ there is absolutely no difference from any other pharmaceutical form. All of the clinical trial requirements for drugs or vaccines apply to medical cannabis; specifically potency, purity, efficacy and identification of side effects to protect patient safety. These can only be ensured through QC testing, supported by rigorous production procedures, as defined by Good Manufacturing Practice (GMP) standards, as laid out in region-specific Pharmacopeial compendia,” says Evolve’s Managing Director, Christopher Busuttil Delbridge.
Sophisticated laboratory methods
The increased need for stringent and compliant quality control and quality assurance in the burgeoning cannabis industry are placing new emphasis on use of sophisticated analytical instrumentation, such as High Performance Liquid Chromatography (HPLC), Gas Chromatography (GC), Mass Spectrometry (MS) and Inductively Coupled Plasma (ICP), all of which require extensive laboratory experience, with training and deep understanding of analytical chemistry.
“The methods and semi-automation of these technologies are coming to the fore. The use of more intuitive software interfaces and a progressively digital-ready workforce lower the threshold of the required learning curve,” Mr. Busuttil Delbridge points out.
“It is evident that the global cannabis production industry mean significant change and challenges must be overcome around analytical testing to provide consistently high-quality products. Scientific consulting firms such as Evolve Ltd have all the required expertise to make this a pain-free experience for anyone investing in this process, especially in the European and sub-Saharan African countries where Evolve operates,” he adds.
About Evolve Ltd
Evolve Ltd, is a Malta-based solution provider and project integrator operating in the European, Mediterranean and sub-Saharan African region. Servicing all scientific workspaces Evolve offers a holistic service that draws on four principal areas of expertise, in the design, equipping, personnel training and maintenance of laboratory and research facilities and equipment.
Evolve Ltd. is part of the Attard & Co. Group of Companies and traces its origins to Attard’s setting up in 1956 of Medical and Scientific Equipment and Supplies (MSES) to cater for its scientific products. During the 1990s, under the leadership of Christopher Busuttil Delbridge, Evolve’s current Managing Director, MSES changed its business model, centered on a vision to become a recognized, award-winning, world-class thought leader and influencer on the international scientific and healthcare laboratory market.
In 2006, following Malta’s accession to the EU, MSES Department was rebranded as Evolve Ltd., an independent Attard subsidiary tasked with serving the scientific community. Within five years, it had developed three operating departments: Medical & Diagnostics, Scientific & Industrial and Technical Services.
Evolve currently employs some 30 people, mostly scientists and technical experts.
For further information, visit: https://evolveltd.eu/
About ATG Pharma
Founded in 2003 by CEO Mike Lio, ATG Pharma (ATG) is a Canadian manufacturer of bench-top equipment designed for dynamic and fast-growing productions. With over 20 years of experience in the pharmaceutical industry, ATG has developed cutting edge benchtop automation technologies for filling and packaging based on deep understanding of market trends and needs. Providing companies with the leading quality standard equipment and service. ATG is proud to provide filling solutions to company’s maximizing their production efficiencies.
For more information, see https://www.atgpharma.com
Resources
Click on Grand View Research Report: Legal Marijuana Market to read report summary.
Click on Ensuring Regulatory Compliance for Medical Cannabis to read original Open Access Government article.
Click on Evolve Newsroom to see further news and events from Evolve Ltd.