By Molnár-Institute for…
DryLab®4 Liquid Chromatography (LC) techniques
Molnár-Institute’s proprietary software suite, DryLab®4, has been developed as an experiment-based universal tool, which works across a very wide range of liquid chromatography (LC) techniques.
These include reverse phase high performance and ultra-high performance liquid chromatography (RP- HPLC/UHPLC), reverse phase with ion pairing (IP-RP), normal phase (NP), hydrophilic interaction (HILIC), ion exchange (IEX) based on salt- or pH-gradients, and gas chromatography (GC).
How DryLab® works
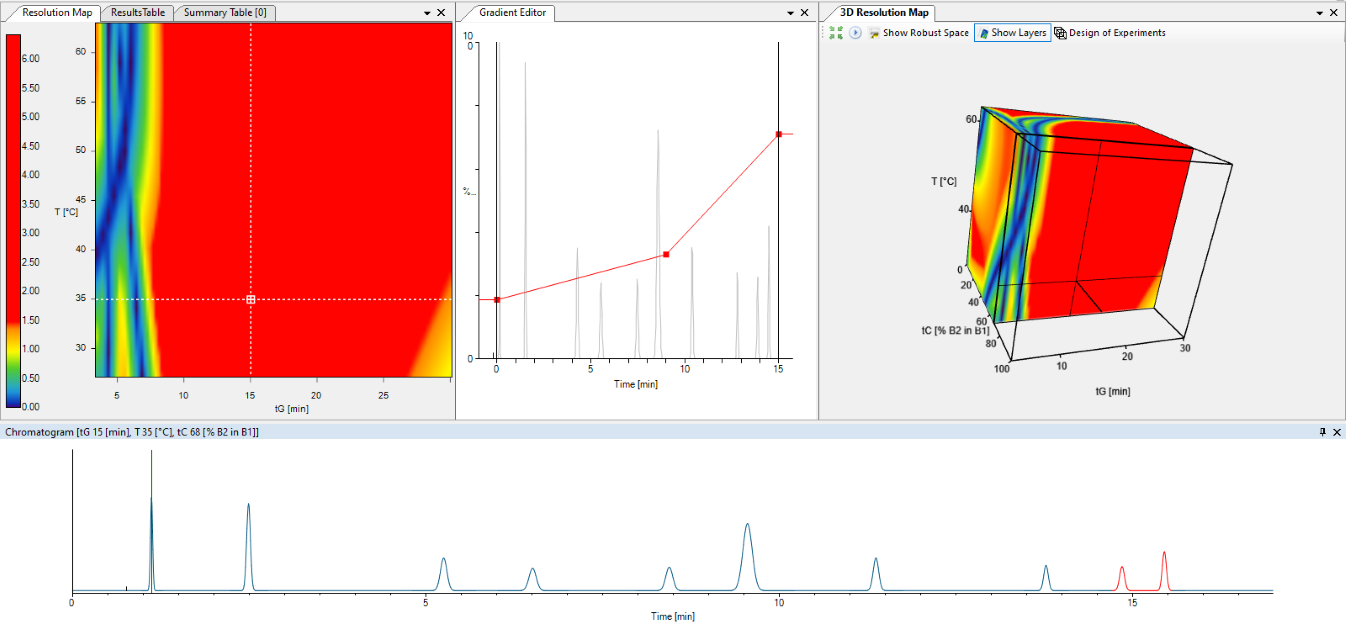
DryLab® can predict resolution and retention times for millions of unique, virtual chromatograms, using data generated from a few input experiments. This provides analytical scientists with the tools needed to achieve a separation and detailed characterization of therapeutic proteins biopolymers and antibody drug conjugates (ADCs) using liquid chromatography.
DryLab® employs a simple step-by-step process that starts with defining an analytical target profile (ATP) for which DryLab® will prompt the researcher to select a suitable workflow and initial method conditions. Data from initial input experiments are then uploaded as universal formatted *.cdf files, for in silico analysis, as well as method validation.
Choice of LC-techniques
DryLab® models chromatographic selectivity; optimizing the resolution between chromatographic peaks that will be quantified using a detector.
DryLab® optimizes band spacing, i.e., the separation, selectivity and critical resolution using first-principle approaches, such as Linear Solvent Strength and Solvophobic Theory.
This works for all non-physical techniques that apply non-covalent interactions between the stationary and mobile phases to separate small or large molecule analytes and can be used across multiple HPLC application studies including pharmaceutical, food, and environmental analysis.
DryLab® methods explored
Applications of DryLab® in various LC techniques have been explored in numerous studies.
These include:
Reverse Phase liquid chromatography (RPLC): A study proposing a generic method development approach for separation of antibody drug conjugate species by RPLC (Fekete et al, 2017) reported a viable generic method development strategy based on optimization of mobile phase temperature, gradient profile and ternary mobile phase composition that achieved fast and efficient separation of the DAR species of a commercial ADC sample, brentuximab vedotin. Based on a limited number of preliminary experiments, a viable method was achieved, based on the observed peak area ratios of RPLC chromatogram of the reduced ADC sample. The prediction offered by DryLab®’s 2D or 3D retention models was found to be highly reliable, with average error of retention time prediction always lower than 0.5%. This was a first presentation of 3D retention modelling for a large therapeutic protein [1].
Hydrophobic interaction chromatography (HIC): a team based at the University of Geneva in Lausanne explored optimization of non-linear gradients in hydrophobic interaction chromatography for the analytical characterization of ADCs (Bobály et. al, 2016) [2]. This team aimed to evaluate the potential of non-linear gradients in HIC, to improve the separation between different homologous species of commercial ADC’s. They measured selectivities between Brentuximab Vedotin species using linear, power function based and logarithmic gradient profiles, discovering that the last one provided the most equidistant retention distribution for the DAR species and best overall separation of cysteine linked ADC in HIC. The study also modelled the logarithmic behavior of DAR species of ADC in HIC using two different approaches, based on both linear solvent strength theory (LSS) and a newly derived equation. In both cases, the retention predictions using two logarithmic scouting gradients were excellent and the error was systematically below 3%, compared to experimental values.
Hydrophilic interaction liquid chromatography (HILIC): Dr. Bobály also led an analysis of a generic method development approach to separation of recombinant mAbs in HILIC using DryLab® separation modelling (Bobály et al., 2017). This study applied the recently introduced widepore stationary phase that expands HILIC applications to larger molecules, such as therapeutic proteins. The study used seven approved mAbs possessing various isoelectric points (pI) and hydrophobicities as well as a cysteine conjugated ADC. Samples were digested by IdeS enzyme, with digests further fragmented by chemical reduction before separation by HILIC, with DryLab® then used to build up retention models. The main benefit of this method was to enable chromatographic separation of polar variants (glycovariants) in a reasonable analysis time at the protein level. Experimental and predicted chromatograms showed good agreement with average retention time prediction error <2%. [3].
Salt-based Ion Exchange separation (IEX): Dr. Szabolcs Fekete, who has been a consistent presenter in all these research studies, led two studies during 2015 that explored method development for the separation of monoclonal antibody charge variants using cation exchange chromatography (CEX). The Part I study, examining a salt gradient approach, aimed to overcome the time intensive and product specific bottlenecks associated with this approach, demonstrating that mobile phase temperature was not relevant for tuning selectivity, while optimal salt gradient program could easily be found, based on only two initial gradients of different slopes and using three initial runs at different pH’s to optimize mobile phase pH. Based on just nine hours of initial experiments to simultaneously optimize salt gradient profiles and pH in IEX, DryLab® was used to treat the data to identify optimal conditions with excellent accuracy of retention times prediction (> 99%). The study showed salt gradient CEX to be a viable generic multi-product approach. [4].
pH-based IEX: In the second part of the study, Dr. Fekete and team evaluated a CEX pH gradient approach for characterization of 10 model mAbs, including panitumumab, natalizumab, cetuximab, bevacizumab, trastuzumab, rituximab, palivizumab, adalimumab, denosumab and ofatumumab. They demonstrated, that retention and resolution could be modelled in this mode, based on only four initial runs (two gradient times and two mobile phase temperatures), requiring only six hours for complete method optimization and again achieving excellent prediction accuracy at around 99%. They concluded that for most compounds, variants were better resolved in the salt gradient mode, where peak capacities were higher [5].
DryLab® for small and large molecule characterization
Dr. Fekete has also collaborated with others to assess applicability of computer-assisted method development and retention modeling to the characterization of both typical small pharmaceutical compounds through impurity profiling and large therapeutic proteins. [6]. His team showed that by choosing appropriate initial conditions, method development for small and large molecule compounds could be performed in less than one day. However, for small molecules possessing different physicochemical properties, the conditions could be very diverse, whereas for biopharmaceuticals such as mAbs or ADCs, a generic method could be more easily developed. The study also showed that DryLab® computer modelling could also be used for simulated robustness testing. Depending on the applied retention model, the impact of any change among six experimental parameters (tG, T, pH, ternary composition, flow rate, and initial- and final mobile phase compositions) on the separation could be assessed using a 26 or 36 type virtual factorial design, with no additional experiments required to perform the robustness evaluation. The team also showed feasibility of virtual method transfer between different chromatographic systems.
Study References
- Fekete, S., Molnár, I., Guillarme, D., (2017) ‘Separation of antibody drug conjugate species by RPLC: A generic method development approach; Journal of Pharmaceutical and Biomedical Analysis, 137, pp. 60-69, https://doi.org/10.1016/j.jpba.2017.01.013
- Bobály, B., Randazzo, G.M., Rudaz, S., Guillarme, D., Fekete, S., (2017), ‘Optimization of non-linear gradient in hydrophobic interaction chromatography for the analytical characterization of antibody-drug conjugates’, Journal of Chromatography, 1481, pp. 82-91, https://doi.org/10.1016/j.chroma.2016.12.047
- Bobály, B., D’Atri, V., Beck, A., Guillarme, D., Fekete, S., (2017), Cusumano, A., Guillarme, D., Beck, A., and Fekete, S. (2016) ‘Analysis of recombinant monoclonal antibodies in hydrophilic interaction chromatography: A generic method development approach’, J Pharm Biomed Anal, 145, pp. 24-32. https://doi.org/10.1016/j.jpba.2017.06.016
- Fekete, S., Beck, A., Fekete, J., Guillarme, D., (2015), ‘Method development for the separation of monoclonal antibody charge variants in cation exchange chromatography, Part I: Salt gradient approach’, J Pharm Biomed Anal, 102, pp. 33-44, https://doi.org/10.1016/j.jpba.2014.08.035
- Fekete, S., Beck, A., Fekete, J., Guillarme, D., (2015), ‘Method development for the separation of monoclonal antibody charge variants in cation exchange chromatography, Part II: pH gradient approach’, J Pharm Biomed Anal, 102, pp. 282-289, http://dx.doi.org/10.1016/j.jpba.2014.09.032
- Fekete, S., Kormány, R., Guillarme, D., (2017), ‘Computer-Assisted Method Development for Small and Large Molecules’, com, Vol. 30, Issue 6, pp. 14–21