By Bachem AG
Chemical glycosylation for homogenous adaptable peptides
Bachem’s chemical glycosylation technology permits synthesis of peptides and proteins with pre-attached sugars, selected from a proprietary library of over 50 specific glycans.
This model of chemical glycosylation delivers significant advantages in markedly improving drug half-life, binding affinity and selectivity properties. Furthermore, compared to recombinant products, the chemically synthesized proteins are more homogeneous.
Glycosylation of peptide drugs also provides a powerful route to optimizing lead candidates. Selective, site-specific chemical glycosylation leads to a homogeneous product with potential for more defined bioactivity compared to heterogeneous products.
Chemical Glycosylation Applications and Features
Chemical glycosylation can improve the pharmacokinetic profile of drugs, allowing the developer to:
- Attach any glycan at the desired position
- Specify the number of attached glycans
- Control the glycan structure
Chemical glycosylation can therefor overcome traditional problems faced in the recombinant production of glycopeptides. These include production of glycopeptides that are too heterogeneous, elaborate and costly purification processes and issues with removal of biological contaminants such as cellular debris or viruses.
In contrast, chemical glycosylation can deliver significant advantages, such as:
- Homogeneous products: Chemical synthesis yields well-defined glycopeptides
- Most chemically synthesized peptide drugs can be easily adapted to glycosylation
- Competitive production costs
Chemical Glycosylation Benefits
In general, selective, site-specific glycosylation leads to a more homogeneous product with potential for more defined bioactivity compared to heterogeneous products. Selective chemical glycosylation can also improve the physicochemical properties of peptides, focusing on one or more of the following properties:
- Binding (Kd)
- Half-life
- Stability
- Homogeneity
Optimizing these properties as a result, this can have positive effects on the pharmacological performance of a lead candidate drug, which can include:
- Improved bioactivity
- Modified receptor selectivity
- Prolonged half-life, providing extended dosing duration
- Better solubility, enabling improved formulation properties
- Improved response to therapy
- Better tolerance of drug
Producing Glycopeptide APIs in Partnership
The partnership between Bachem and GlyTech is focused on the chemical development and manufacturing of glycosylated peptides.
Bachem has the proven expertise to scale up and manufacture kilogram scale peptides, while GlyTech is capable of producing glycans in kg amounts by a proprietary technology.
“The selective chemical glycosylation at large scale has potential to be applied to a variety of peptides, where we can pioneer the concept of improving current and future drugs via chemical glycosylation,” explains Bachem CMO, Dr. José de Chastonay.
Bachem’s goal is to improve physicochemical properties of peptides through selective chemical glycosylation. This can be achieved thanks to three key advantages.
- Both chemical structure and position of glycosyl moieties can markedly improve the biological activity of a peptide.
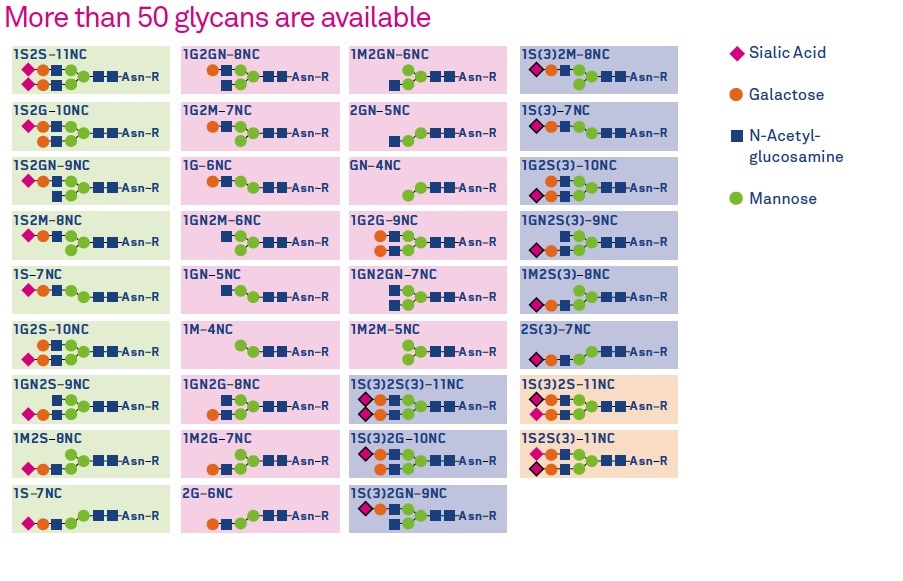
- A comprehensive library of glycans, containing human glycoproteins linked to asparagine, has been created.
- Well-defined glycosylated structures can be generated through chemical synthesis, which allows optimal choice of the glycosyl moiety and its position.
The chemical glycosylation of Interferon β-1a provides a powerful proof of concept.
Bachem and GlyTech shared the 2013 CPhI innovation prize finalist position for their groundbreaking work on Interferon β-1a. Since then, the technology has been used in multiple other projects, including the manufacture of glycosylated somatostatin analogues and glycosylated GLP-1. In all cases, drug improvements were achieved.
Selective Glycosylation Services
Generating a library of glycosylated analogs can be a powerful way to enable optimization of lead candidates. This is achieved by a “library” approach:
- For a lead peptide, which has been chosen for glycosylation, a human type glycan library is available.
- The optimized glycopeptide designs will be selected in a joint project team of the client together with Bachem and GlyTech, to benefit from the respective expertise.
- Synthetically glycosylated peptides will be produced in small scale (mg-g) by GlyTech.
- The glycosylated peptides will be screened by the client in order to find a glycopeptide with improved pharmacological properties.
- A glycopeptide with enhanced properties can then be scaled- up and manufactured by Bachem in the required quantities.
More than 50 glycans are available, based on various permutations of Sialic Acid, Galactose, N-Acetyl-glucosamine and Mannose.
A typical project outline follows a five-phase process:
Lead optimization phase: Asses and define current compound properties, identify desired properties
Design phase: Scientific dialogue and collaboration between partner and Bachem/GlyTech to design a number of compounds expected to meet desired properties
Synthesis & supply phase: Synthesize compounds at milligram or gram scale for research purpose (non-GMP), supply compounds with agreed sequence, structure and quantity
Test phase: Positive results may lead to a License Agreement or subsequent iteration of Design Phase for further improvement of lead compound
Scale-up phase: Scale-up and analytical development at Bachem with cGMP manufacturing
Resources
Click on Bachem Chemical Glycosylation of Peptides for more product information.
Click on Bachems News to see latest News & Events.