By Brinter Ltd.
Brinter® 3D bioprinter used to manufacture GBP gabapentin tablets for enhanced pet safety
Turku, Finland: – The pioneering modular multi-material 3D bioprinting platform Brinter® has enabled a Finnish-based academic research team to produce precise tablet veterinary dosage forms of the anti-convulsant gabapentin (GBP), potentially filling a serious gap in the market.
Currently, there are few to no gabapentin tablets available in veterinary markets, leading to pets being treated with compounded dosage GBP forms or off-label use of human tablets being sub-divided ‘by eye’, raising significant risks of under- or over-dosing.
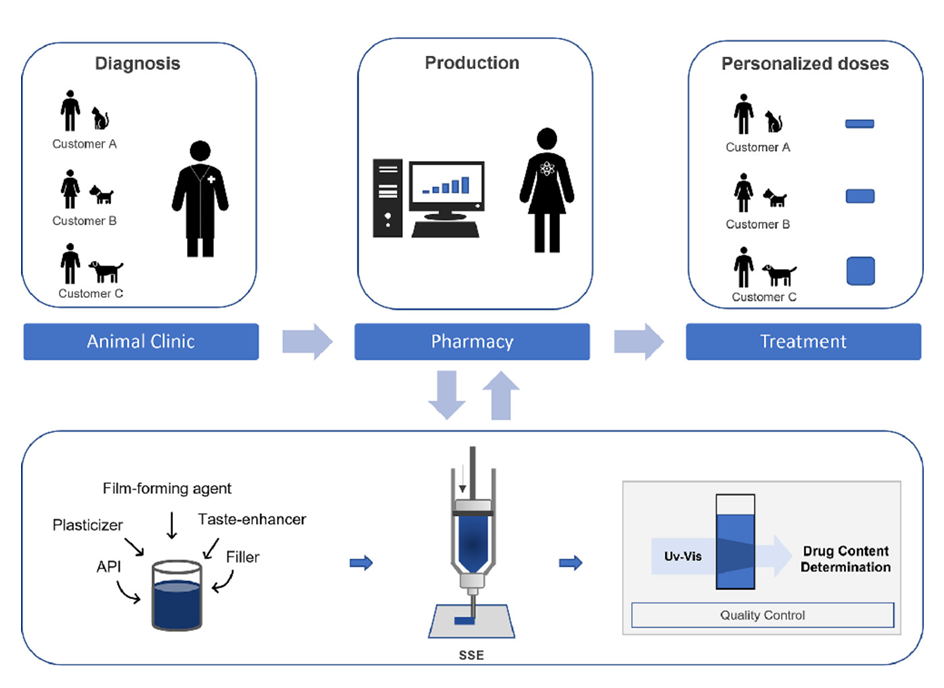
A team headed by Dr. Erica Sjöholm from the Pharmaceutical Sciences Laboratory at the Faculty of Science and Engineering of Åbo Akademi University in Turku, Finland, set out to find a more precise method to quantify the challenging gabapentin small molecule, which lacks chromophores. Their second aim was to develop a new manufacturing technique to accurately and rapidly prepare veterinary GBP dosage forms that could be used close to point-of-care.
SSE 3D printing
Their study ‘Semi-solid extrusion 3D printing of tailored ChewTs for veterinary use – A focus on spectrophotometric quantification of gabapentin’ published in the European Journal of Pharmaceutical Sciences shows that semi-solid extrusion 3D printing is a feasible way to manufacture pet-friendly tailored gabapentin dosage forms. The team also succeeded in developing a new method to analyze GBP using UV–Vis spectrophotometry via an ascorbic acid derivatization method.
For GBP chewable tablet production, the Åbo Akademi team developed a semi-solid extrusion (SSE) 3D printing material extrusion technique that deposits a gel or a paste according to a pre-determined design. They used the Brinter® One 3D BioPrinter fitted with the Pneuma Tool print head, which enables low to medium viscosity inks, pastes, or gels to be dispensed with the aid of compressed air. The team also used the browser-based Brinter® printing software to control layer height, shell, fill and angle settings, along with print speed and dispensing pressure to achieve therapeutic doses ranging from 10 mg to 200 mg.
Tailored doses
Using this set-up, the team found that a set of eight 10 mg GBP 3D printed tablets (printlets) could be produced in just over two minutes, with the potential to accelerate this further by increasing print speed and air pressure. They also observed that the average wet weight of the printlets correlated very closely to theoretical design size and dry weight (R2 = 0.9966), making SSE 3D printing highly suitable for preparing individually tailored dosage forms. The printed GBP tablets were given a brown color and liver scent to make them attractive to cats and dogs.
The research team say their study findings open the door to many practical benefits in showing that pet-friendly GBP dosage forms can easily be manufactured and analyzed.
“The UV–Vis quantification method with AA derivatization is simple and can fairly easily be implemented in pharmacies, veterinary clinics, animal hospitals, and such. The suggested chewable formulation of GBP serves as an example of a dosage form that is simple to prepare and enables tailoring of the dose. Implementing these findings in practice could diminish the current need for extensive manual labour when compounding GBP or other drug-loaded dosage forms or the risk associated with the splitting of tablets and capsules. Instead, safe and effective veterinary medicines could be rapidly manufactured at or close to the point-of-care,” the study concludes.
About Brinter®
Founded in 2020 via partial demerger in Turku, Finland, Brinter® is the manufacturer of a modular multi-material bioprinting platform scalable from manual R&D to automated production. The company works with science and bioengineering organizations to integrate 3D bioprinting into their research, manufacturing, and treatment methods. The goal is to break through current technological limitations and improve people’s quality of life.
Brinter® has bootstrapped its growth since 2019, when it launched its first product, and is currently active in more than ten countries, including Germany and the UK.
Customers of the company now include bio and pharmaceutical companies like Nanoform, as well as research organizations like VTT, BEST group at the University of Glasgow, Johannes Gutenberg University of Mainz, the University of Oulu, and the University of Helsinki, whose directors are enthusiastic about the technology’s potential.
To date, Brinter® has raised a total of €1.2M in funding and is currently active in more than ten countries, including the USA, Germany, India, and the UK.
For more information, visit www.brinter.com.
Resources
Click on Semi-solid extrusion 3D printing of tailored pharmaceutical ChewTs for veterinary use – A focus on spectrophotometric quantification of gabapentin to read the full study.
Click on Brinter – Easy to use to see a video of Brinter® One in action