By Brevetti Angela…
Brevetti Angela and 3CK are stars in Universe of Pre-Filled Syringes Conference
Vicenza, Italy: – Leading manufacturer of Blow-Fill-Seal (BFS) pharma packaging machinery, Brevetti Angela Srl , and its sister company 3CK are prominent presences at the 2022 PDA Universe of Pre-Filled Syringes and Injection Devices Conference in Palm Springs, California.
Brevetti Angela has been a consistent presence at PDA-organised events for more than a decade, underlining its status as a leading force in parenteral pharma packaging. It is now joined by 3CK as an innovator in the specific field of pre-filled and pre-fillable plastic syringes.
New pre-filled O-Flow concept
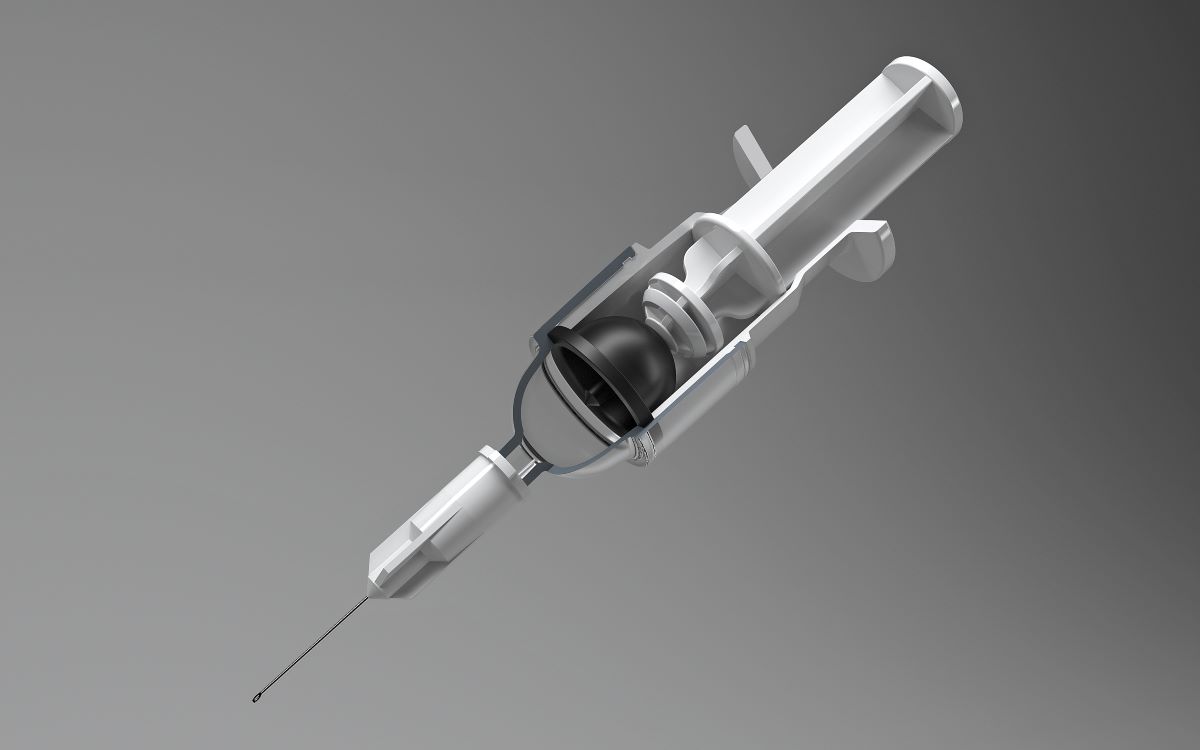
Jointly exhibiting from Booth 504 at the Palm Springs Convention Center, the two companies are focusing on the latest research on injection devices. Coming up from years of research within the BFS technology, the latest generation of prefilled syringes presented at the booth focuses on the highly innovative 3CK O-Flow pre-fillable syringe, which will be available for display during the show.
The fifth generation O-Flow silicone-free syringe concept was jointly developed, based on Brevetti Angela’s long experience in applying BFS technology to parenteral packaging, including the patenting of SECUREJECT® as the world’s first machine to apply Blow-Fill-Seal technology to production of pre-filled syringes.
Silicone-free
O-Flow takes this a step further with a single use prefilled plastic syringe that is safe and cost-effective to manufacture, while at the same time being easy and safe to use. Its ground-breaking design can be adapted to volumes up to 20ml, needs no silicone oil and eliminates break loose force, making it exceptionally user-friendly and convenient to produce.
O-Flow is produced and marketed by 3CK while Brevetti Angela has also developed the BFS O-Fill aseptic filling system, specifically designed to accommodate the 3CK format.
About Brevetti Angela
Brevetti Angela Srl is one of the world’s leading manufacturers of pharma primary packaging machinery, specializing in production of Blow Fill Seal (BFS) machinery for the aseptic packing of liquids in purpose formed plastic bottles, vials and parenteral forms. Brevetti Angela platforms are ideally suited to aseptic filling of pharmaceutical solutions and can also be applied to food & beverages, cosmetics or cyanoacrylates filling.
Founded by Francesco Consolaro in 1977, the company remains based in Arzignano, in the province of Vicenza in northeastern Italy. Over more than four decades, Brevetti Angela has been guided by a vision of following a path of continuous innovation based on ensuring customer satisfaction and quality of finished product, with a mission to provide primary packaging solutions that are flexible, versatile and also reliable and easy to maintain.
The company is particularly known for its range of advanced and patented SYFPAC® BFS machines designed and engineered to achieve the highest quality aseptic packaging of sterile pharmaceutical fluids for small volume vials, bottles, and pre-filled syringes.
One of the key enablers for very high quality at Brevetti Angela is its emphasis on keeping all vital processes ‘in house’ at Arzignano where all facilities meet CE standards and since 2008 have been certificated to ISO 9001:2008 quality standards.
About 2022 PDA Universe Conference
Building on the success of past US Parenteral Drug Association events, the 2022 PDA Universe of Pre-Filled Syringes and Injection Devices Conference focuses on celebrating the latest advances in pre-fillable syringe technology and applications.
Themed ‘The New Normal in Injectable Drug Delivery’, the Universe of Pre-Filled Syringes conference is a two-day event opening October 18 at the Palm Springs Convention Center in southern California.
The conference is exploring diverse aspects of the future of pharmaceutical parenteral devices, focusing on technological advances that will push the industry ahead with a wide array of industry experts sharing experiences, new developments, regulatory considerations, challenges, and industry trends.
It will explore continued advances in materials of construction, manufacturing processes, injection processes and safety devices, and other technology improvements that create a dynamic environment in the application of pre-filled syringes or other pre-filled containers and injection devices to drug delivery.
The event is organized by the PDA Parenteral Drug Association with further information at https://www.pda.org/global-event-calendar/event-detail/2022-pda-universe-of-pre-filled-syringes-and-injection-devices-conference
Resources
Click on O-Flow: The Next Generation of Pre-Filled Syringes for further information from 3CK.