By Bachem AG
Bachem White Paper analyses ‘back to roots’ return to stirred-bed vessels for oligomers
Bubendorf, Switzerland: –Peptide and oligomer technology specialist Bachem has authored a White Paper presenting rediscovery of traditional stirred-bed vessel processes as a modern solution for industrial scale production of oligonucleotides or oligos.
The technical paper shows how returning to stirred-bed vessel methodology is an innovative way of meeting the challenge of rising industry demand for oligomers at commercial scale to returning to the roots of peptide synthesis while introducing leading-edge chemical and engineering adaptions that have succeeded in raising concentration levels to an unprecedented two moles of oligonucleotides per batch – corresponding to a double digit-kilogram-scale for an average oligomer.
Need for new SPOS solution
Stirred bed vessels were used in pioneering DNA synthesis processes in the 1980s. In the last decades, however, oligonucleotide manufacturers prevailed solid-phase oligonucleotide synthesis (SPOS) in fixed-bed flow through column systems as industry standards. These flow-through oligonucleotide synthesizers allowed better automation, fast chemistry, and low cycle times.
Large-scale solid phase oligonucleotide synthesis (SPOS) production of oligo-based active pharmaceutical ingredients (APIs) is needed more urgently than ever, with hundreds of oligomer-based drugs in advanced clinical trials and potential metric ton demand.
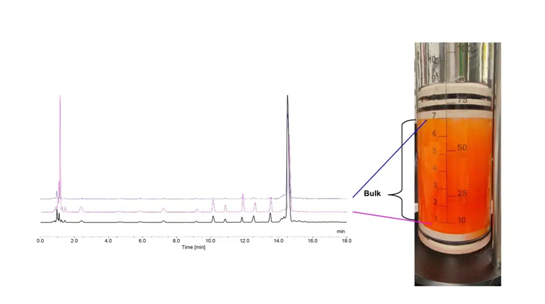
However, the scalability of the manufacturing using flow-through synthesizers is limited by factors such as pump size in generating flow or thickness of the solid-phase resin bed height. The latter is particularly critical in being able to ensure efficient coupling reactions.
A further important disadvantage of flow-through technology at all batch sizes is concentration gradient over the column (Figure 1), resulting in batch inhomogeneity. Better batch homogeneity can be achieved by moving away from flow-through column principles to agitated-bed systems. This inhomogeneity of conventional packed bed flow SPOS is completely omitted in the stirred-bed technology (SBT) SPOS due to uniform reagent contact time and distribution.
Back to the Future
When Bachem scientists started the journey back to stirred vessels, they wanted to demonstrate the feasibility of scale-up to industrial scale, establishing a toolbox and generic process parameters that apply to most oligonucleotides.
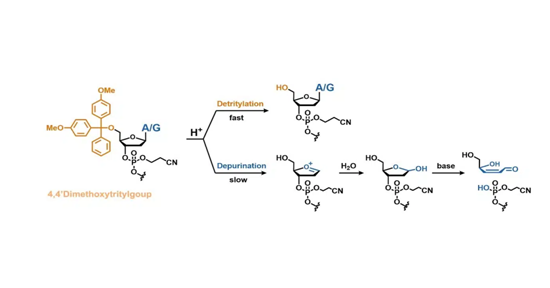
One milestone along the way was solving the problem of depurination, the side reaction from acidic conditions during detritylation removal of the protection groups in SPOS (Figure 2). This decreases yields and purity more markedly in stirred vessels than in flow-through columns.
Extensive testing of different reagents, acids, and scavengers has found new deprotection protocols to suppress this side reaction, replacing the common dichloroacetic acid and toluene cocktail during deprotection with an acid with a higher pKA value, a mixture of toluene with trifluoroethanol and special scavengers.
Bachem has now conducted more than 500 lab-scale SPOS processes applying SBT to make oligonucleotide-based APIs, with several case studies performed at a multi-gram scale with “real” therapeutic oligonucleotide sequences.
SBT SPOS for Large-Scale Oligomer Production
These have revealed that SBT is not only suitable for making full-length product sequences but is also ideal for hybrid approaches: short fragment synthesis with typically less than 10 nucleotides at the hundred-kilogram scale followed by either chemical or enzymatic ligations with no chromatography steps involved. With SBT, the use of greener and more sustainable solvents is possible and the subject of intense investigations at Bachem.
Combining highly efficient synthesis of oligonucleotides with the highly effective purification by multicolumn countercurrent solvent gradient purification (MCSGP) systems1, in which Bachem has become a pioneering CDMO, has resulted in a further reduction of solvents and improvements of the process mass intensity.
SBT Benefits
Thus, SBT has now been reinvented as a highly efficient process for industrial scale oligonucleotide synthesis, with benefits that include:
- Batch/product homogeneity
- Swelling of solid support resin acceptable
- Application of high-loading solid-support resin feasible
- Seamless scalability
- On-resin modifications possible
- Potential to save solvents and reduce process mass intensity (PMI)
PMI is a key performance indicator for solvent consumption. With SBT, a drastic reduction of solvent consumption and the PMI can be achieved by the higher loading of the solid phase resin and the reduction of solvents used for washing.
Thus innovative SBT for SPOS now represents an economical engineering solution with a simple reactor design and adapted chemistry enabling ton-scale commercial oligonucleotide API manufacture with unmatched process mass intensity.
Reference:
- White paper from Bachem: Continuous Chromatography – Pushing the boundaries of peptide and oligonucleotide production
About Bachem
Bachem is a leading, innovation-driven company specializing in the development and manufacture of peptides and oligonucleotides. The company, which has over 50 years of experience and expertise, provides products for research, clinical development, and commercial application to pharmaceutical and biotechnology companies worldwide and offers a comprehensive range of services.
Bachem operates internationally with headquarters in Switzerland and locations in Europe, the US and Asia. The company is listed on the SIX Swiss Exchange.
For further information, see www.bachem.com.
Resources
Click on Manufacturing Oligonucleotides with Stirred-bed Technology to access Bachem White Paper.
Click on Bachems News to see latest News & Events.