By Bachem AG
Bachem research grade peptides play key roles in assessing ACE2-mediated COVID-19 infection
Bubendorf, Switzerland: –Bachem AG’s research grade peptides, particularly DX600, are playing important parts in new investigations into the controversial role of ACE-boosting drugs in Covid-19 illnesses and alternative treatments.
It is known that Severe Acute Respiratory Syndrome Coronaviruses 2 (SARS-CoV-2), including Covid-19, enter human cells via the external part of the angiotensin-converting enzyme 2 (ACE2) receptor protein. ACE2 is found within the membranes of many cells including the nasopharynx, nasal and oral mucosa, as well as the epithelial cells of lung alveoli, small intestine, colon, kidney, liver, pancreas and vascular endothelium.
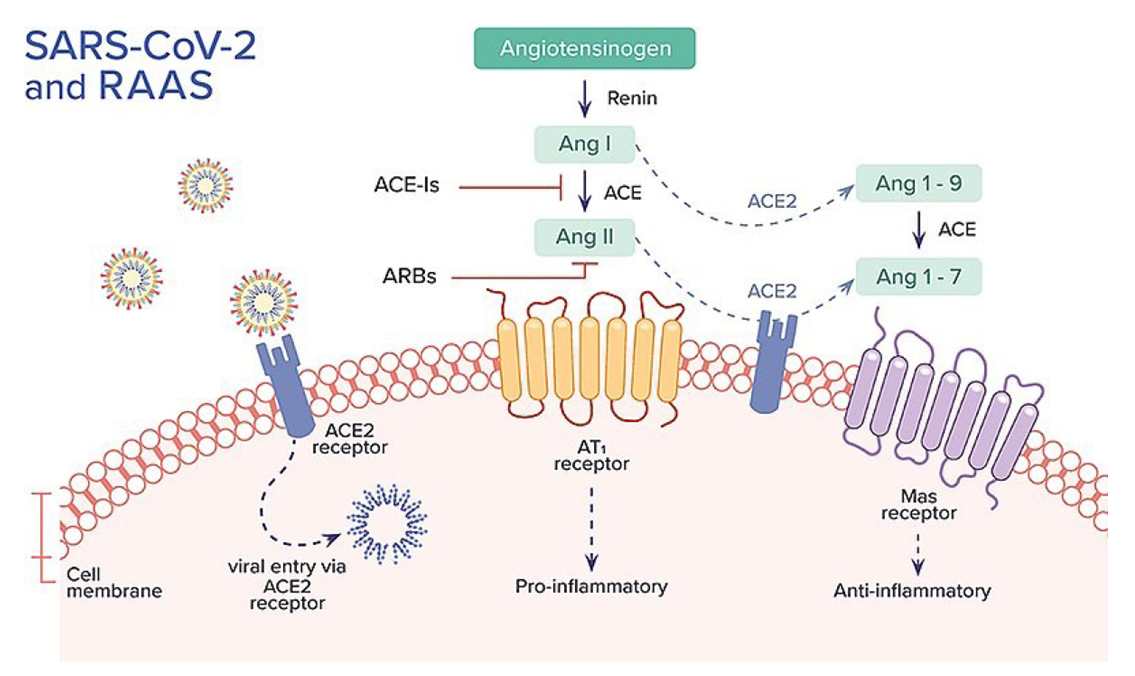
Interaction of SARS-CoV-2 with RAAS
The Renin-Angiotensin-Aldosterone System (RAAS) is an important hormone system for regulating blood pressure and electrolyte balance1, as well as playing a major role for several pathological processes like the acute respiratory distress syndrome (ARDS), fibrosis, hypertension and cardiac dysfunction, whose sufferers also face more severe outcomes from COVID-19 infection. Thus there have been concerns that typical drugs for these patients which increase the ACE2 levels − like the Angiotensin-II blockers (ARBs) and Angiotensin-Converting Enzyme inhibitors (ACEi) − make such patients more susceptible to SARS-CoV-2 infections.
There is considerable scientific uncertainty on this, with an alternative school of thought that ACE boosting drugs on may actually be beneficial in treating COVID-19, since ACE2 is considered to be a protective agent for inflammation, as it increases the level of the anti-inflammatory peptide Angiotensin 1-7 (Ang 1-7) and decreases the level of the pro-inflammatory vasoconstrictor peptide Angiotensin II (Ang II). To be more precise: the membrane bound receptor ACE2 converts the octapeptide Ang II directly to the anti-inflammatory Ang 1-7 and the decapeptide Angiotensin I to Angiotensin 1-9 (Ang 1-9), which can be converted subsequently by the Angiotensin-Converting Enzyme (ACE) to Ang 1-7. The peptide Ang 1-7 then binds to the mitochondrial assembly receptor (MasR) on cell surfaces to stimulate vasodilation and anti-inflammatory effects (Figure 1).
Beneficial RAAS balancers?
In March 2020, researchers from China first reported on higher concentrations of circulating Ang II in plasma samples from COVID-19 patients using commercial ELISA tests. The higher Ang II levels in COVID-19 patients strongly correlated with the viral load and symptoms of lung injury. They suggested that the imbalanced RAAS in COVID-19 patients was caused by SARS-CoV-2 and further argued that RAAS balancing drugs such as ARBs and ACEi might be beneficial to these patients.
Several studies followed showing harmful, beneficial or no effects on the severity of COVID-19 in patients treated with RAAS inhibitors. Then researchers investigated the continuation vs. the discontinuation of treatment with ACEis and ARBs in COVID-19 patients with hypertension and concluded that their use should be continued. Therefore, American and European medical societies currently recommend physicians do not change the usual ARBs and ACEi therapies in COVID-19 patients 1,4. However, the life sciences community agrees that further research is needed and this, in turn, requires an accurate assessment of the angiotensins.
Assessing Angiotensins using Bachem’s Research Grade Peptides
The interaction of SARS-CoV-2 with ACE2 is important due to the therapy decisions that might follow the measurement of the angiotensins in COVID-19 patients. Scientists at the USA’s Hypertension and Vascular Research Center at the Wake Forest University School of Medicine have therefore compared results obtained with commercial ELISA tests with those obtained from a validated radioimmunoassay using the Ang 1-7 and Ang II standards from Bachem in Torrance, CA3. The sensitivity and relative specificity of radioimmunoassays are required to quantify Ang 1-7 and Ang II peptides in our body , whose levels are in measured in picograms per milliliter plasma or per gram tissue. The results of these investigations published in January 2021 concluded ELISAs were largely unable to recognize the Bachem Ang (1–7) and Ang II standards, either because the sensitivities of the ELISAs fell below those stated by manufacturer or inability of the assays to detect authentic Ang (1–7) and Ang II. This may also explain the failure of both ELISAs to detect Ang (1–7) and Ang II in the extracted plasma samples.
The research team recommended that assessment should be made on plasma rather than serum samples with reference standards validated even for commercial clinical testing kits.
Inhibiting SARS-CoV-2-ACE2 Interaction
One possible therapeutic approach for COVID-19 infection would be the blocking of the SARS-CoV-2-ACE2 interaction either with antibodies or some other small peptide molecules4. Scientists at the Cambridge University in the UK recently used beating stem cell-derived heart cells infected with a SARS-CoV-2 spike-pseudotyped virus system to screen potential inhibitors of a SARS-CoV-2 infection. Amongst the test compounds there was the ACE2 peptide antagonist DX600, listed along with the angiotensin peptides used at Wake Forest University in Bachem’s catalog for cardiovascular system and disease applications.
The Cambridge researchers demonstrated that within this screening approach DX600 could “prevent SARS-CoV-2 from entering the heart cells.”5 Furthermore, they found the synthetic peptide up to seven times more effective than the antibody against ACE2 that was also investigated.
The Cambridge team state in their published article, “DX600 is a highly selective peptide that has not been tested previously as a viral entry inhibitor. It forms multiple interactions with the catalytic site of the ACE2 receptor that is distinct from the receptor-binding domain of the virus.”
Their conclusion is that binding of DX600 may be sufficient to change the conformation of ACE2 in such a way that the virus cannot interact with the receptor and enter the cells anymore. There is hope now that these promising results in cells will turn into an effective treatment for COVID-19 patients, with global implications.
References:
- 1Chams, Nour, Sana Chams, Reina Badran, et al. “COVID-19: A Multidisciplinary Review” Frontiers in Public Health 8 (2020): 383
- 2 Liu, Yingxia, Yang Yang, Cong Zhang, et al. “Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury.” Science China Life Sciences, 63 (2020): 364–374
- 3 Chappel, Mark C., Nancy T. Pirro, Andrew M. South, TanYa M. Gwathmey. “Concerns on the Specificity of Commercial ELISAs for the Measurement of Angiotensin (1-7) and Angiotensin II in Human Plasma” Hypertension. 77, 3 (2021): e29-e31
- 4 Kumar Shuklar, Ashwin, and Monisha Banerjee. “Angiotensin-Converting-Enzyme 2 and Renin-Angiotensin System Inhibitors in COVID-19: An Update.” High Blood Pressure & Cardiovascular Prevention 28, 2 (2021): 129-139
- 5 Williams, Thomas L., Maria T. Colzani, Robyn G. C. Macrae, et al. “Human embryonic stem cell-derived cardiomyocyte platform screen inhibitors of SARS-CoV-2 infection.” Communications Biology 4, 926 (2021)
About Bachem
Bachem is a leading, innovation-driven company specializing in the development and manufacture of peptides and oligonucleotides.
With 50 years of experience and expertise Bachem provides products for research, clinical development and commercial application to pharmaceutical and biotechnology companies worldwide and offers a comprehensive range of services.
Bachem operates internationally with headquarters in Switzerland and locations in Europe, the US and Asia.
The company is listed on the SIX Swiss Exchange. For further information, visit the Bachem website.
Click Here for more information on Bachem’s products.
Resources
Click on Assessing and Influencing ACE2-mediated COVID-19 Infection to read original Bachem News item.
Click on Bachems News to see latest News & Events.