By Bachem AG
Bachem ramps up oligonucleotide manufacturing to meet anticipated mass demand
Bubendorf, Switzerland: – World-leading CDMO partner Bachem AG observes that oligonucleotides are moving beyond advanced drug modalities for tackling rare diseases to show potential to power new mass market drugs that combat common diseases affecting huge numbers of patients worldwide.
Accordingly, Bachem has underlined its prowess and agility as a leading CDMO provider of peptides and oligonucleotides by developing new oligomer chemistry and manufacturing capabilities to meet these anticipated future market demands for oligonucleotides at unprecedented scale.
Excited about oligos
Bachem’s Head of Oligonucleotides, Dr. Daniel Samson, says oligomers are rapidly moving beyond ‘niche’ status for the pharma industry.
“By nature, rare diseases were the first targets, but we see more and more trends towards larger indications, larger populations. This generates a lot more demand, and more capacity is needed. In addition, big pharma companies are getting more and more excited. So, that’s certainly a trend. We’ll have both rare diseases of small indications, but also large indications,” says Dr. Samson, adding that this will require an entirely new level of manufacturing capability.
“When it goes to later clinical phases or even commercial demand for large indications, we talk almost about metric tons of one single molecule and that’s a completely different story than in terms of equipment, trains, how to manufacture that,” he explains.
Meeting supply and capacity challenges
Bachem was early to recognize the opportunity to expand its existing manufacturing capacity and expertise in large-scale active pharmaceutical ingredients (APIs) by building facilities to produce oligonucleotide APIs. However, this change in scale and high demand raises new challenges related to capacity, sustainability, supply chain, cost, and quality.
The Swiss-based company is meeting these challenges with investments in new facilities, people, and innovations. Building on its long-established expertise in peptide drug development and commercialization, Bachem has been able to apply synergies in technology and know-how to oligomer manufacturing.
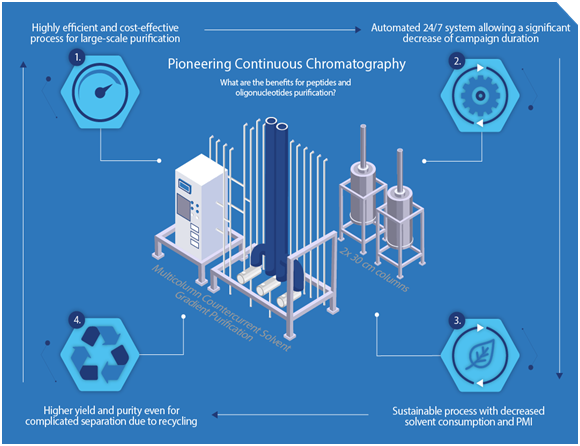
As a first mover in the industry, Bachem is pioneering the implementation of automation, new methods, and technologies for synthesis and purification, such as continuous chromatography on a commercial scale.
Multiple level program
Daniel Samson says this has required Bachem to respond on multiple levels: human, infrastructure and process development.
“We are hiring a lot of people and we train them. We are operational for quite a while now and have already manufactured several oligonucleotides for our customers,” he says.
“For the mid and longer term, we invest a lot of CapEx (capital expenditure) in the construction of a new building, new equipment, and innovation as well,” Dr. Samson notes.
“Innovation helps, especially for larger scale, in decreasing the process mass intensity, the carbon footprint, and the solvent consumption,” he adds, pointing to examples such as implementation of continuous chromatography, as “absolutely one of the methods of choice to save solvent and at the same time have a very powerful separation.”
Long-term ambition
Dr. Samson says Bachem’s goal is to become one of the leading CDMO for peptides and oligonucleotides by offering customers a full package of end-to-end services for oligo-based compounds, from process development and analytics to regulatory affairs and commercialization.
“We are ready to grow. We are investing heavily in a new facility building at Bubendorf and an additional site at Sisslerfeld. We will provide the capacity and at least some of the oligonucleotides will go very big. So, that’s our ambition to provide those,” Dr. Samson declares.
About Bachem
Bachem is a leading, innovation-driven company specializing in the development and manufacture of peptides and oligonucleotides. The company, which has over 50 years of experience and expertise, provides products for research, clinical development, and commercial application to pharmaceutical and biotechnology companies worldwide and offers a comprehensive range of services.
Bachem operates internationally with headquarters in Switzerland and locations in Europe, the US and Asia. The company is listed on the SIX Swiss Exchange.
For further information, see www.bachem.com.
Resources
Click on Oligonucleotide NCEs for further information on Bachem oligomer capabilities.
Click on Innovation transforming Oligonucleotide Manufacturing to learn more.
Click on Bachem continuous chromatography to watch webinar.
Click on Bachems News to see latest News & Events.