By Bachem AG
Bachem multi-column purification overcomes barrier to large scale ‘tide’ production
Bubendorf, Switzerland: –The leading CDMO Bachem has taken a major step to enabling sustainable large-scale production of valuable therapeutic peptide and oligonucleotide APIs with a revolutionary new purification technology.
Bachem’s incorporation of multi-column countercurrent solvent gradient purification (MCSGP) into its downstream process allows peptides to be produced at the commercial scales demanded by the pharma industry without the previous need for huge quantities of chemical solvents with their problems of pollution and waste.
Mass market peptides
The recent and continuing growth in the peptide drug market has been supported by a strong pipeline of peptide drugs in development, with many expected to be approved for use in the coming years. This growing pipeline is placing ever-higher demands on manufacturing capacity for peptide and oligomer-based active pharmaceutical ingredients (APIs), especially with the advent of oral and inhaled peptide products. Pharma companies are now under pressure to be able to generate several hundred kilograms (kg) of peptide active pharmaceutical ingredients (APIs) per year.
The need is not only to manufacture a greater range of ‘tide products but also to do so at larger scale to make them commercially viable. Between 2018 and 2020, the average peptide manufacturing batch size more than doubled in response to a wider variety of peptide drug modalities entering the market. The need for large-scale manufacturing of peptide APIs has been further increased with the advent of new GLP-1 analogs that target metabolic diseases like diabetes and obesity in huge patient populations.
Search for sustainability
A significant challenge currently standing in the way of large-scale peptide production lies in the complex downstream purification process and the chemical solvents involved, which go counter to the other current industry imperatives for sustainable processes and environmental responsibility.
For example, when producing the glucagon-like peptides (GLPs) that are often used in treatments for diabetes and obesity, producing each kilogram of API could involve up to 16,000 kg of chemicals, many of them solvents that are problematic to filter and dispose safely.
Peptide manufacturing processes in general use excessive amounts of solvents and produce considerable quantities of waste, especially the purification step.
Therefore, pharmaceutical companies look for a manufacturing partner that can process large-scale demand of a peptide API while meeting the three key commercial demands:
- Reducing environmental footprint with minimized solvent consumption and waste
- Maximizing throughput and productivity to speed up manufacturing processes
- Increased yield at target purity
Continuous chromatography
One of the most critical aspects of peptide production is purification, to ensure the final product is free from impurities and contaminants. The most efficient approach to peptide purification is chromatography.
This method involves passing the peptide solution through a column filled with a stationary phase, typically silica or beads, and an elution solvent, such as water or buffer. The peptides will bind to the stationary phase, while the impurities will pass through. Chromatography is a highly efficient and selective method of purification, but it can generate a lot of waste and material consumption at large-scale as well as long operation-time.
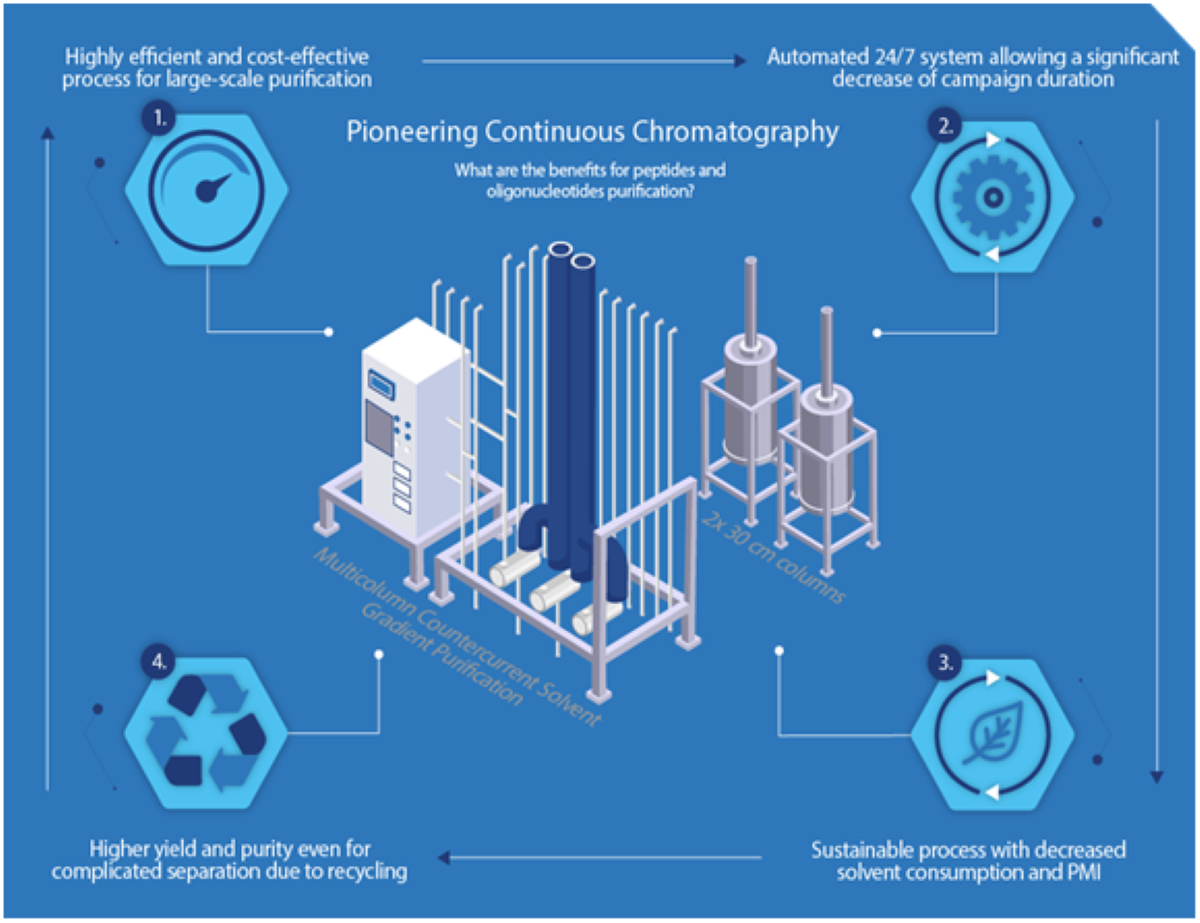
The Bachem solution is based on two elements working in synergy.
The first is continuous chromatography that can operate 24/7 through an automated system, reducing purification cycle times. A continuous process is inherently a more sustainable process, reducing solvent consumption and process mass intensity. For large-scale manufacturing, continuous processes offer particularly effective solutions to the challenges of maximizing yield and quality while ensuring sustainability. Accordingly, Bachem has prioritized continuous chromatography as a promising purification solution due to its high productivity, improved selectivity, and reduced costs.
MCSGP advantages
The second element is the world’s first Multicolumn Countercurrent Solvent Gradient Purification (MCSGP) system for center-cut purification on an industrial scale. This technology represents a major advance in the downstream processing of oligonucleotides. It typically results in higher capacity with lower solvent consumption and higher yield compared to conventional single-column batch purification.
Bachem acquired its first two process-scale MCSGP systems with accompanying control strategies and workflows in 2021 for the purification of peptides and oligonucleotides. Its systems are qualified for GMP use and have been used to that level for purifications at industrial scale.
MCSGP is a scalable, cost-effective, and highly efficient process, particularly for large-scale production, as the use of standard chromatographic conditions in MCSGP does not affect the quality of the API, making it a technology that improves the capacity, quality, and sustainability of the purification process.
With this innovative technology in hand, waste, solvents, and operation time are drastically reduced while throughput and productivity are significantly increased.
About Bachem
Bachem is a leading, innovation-driven company specializing in the development and manufacture of peptides and oligonucleotides. The company, which has over 50 years of experience and expertise, provides products for research, clinical development, and commercial application to pharmaceutical and biotechnology companies worldwide and offers a comprehensive range of services.
Bachem operates internationally with headquarters in Switzerland and locations in Europe, the US and Asia. The company is listed on the SIX Swiss Exchange.
For further information, see www.bachem.com.
Resources
Click on Greener Purification at Large-Scale to access downloaded case study.
Click on Bachem for further information.
Click on Bachems News to see latest News & Events.