By Skyepharma Production…
Skyepharma develops NanoMicS nanoencapsulation platform to usher in new era for OSD forms
Saint-Quentin-Fallavier, France: – Researchers from oral dosage specialist CDMO Skyepharma Productions S.A.S. (Skyepharma) and Lyon University have authored a study detailing how their jointly developed NanoMicS platform delivers significantly enhanced performance in the oral administration of nanoparticles.
Dr. Oksana Lemasson, and Dr. Sandrine Bourgeois, from the Laboratory of Automatic Control, Chemical and Pharmaceutical Engineering (LAGEPP) at Claude Bernard University Lyon, and Dr. Vanessa Bourgeaux, Innovation Leader at Skyepharma Production, discuss the performance of the NanoMicS platform and its potential applications.
Overcoming Bioavailability Barriers
Their article, published in the online magazine www.ondrugdelivery.com, discusses the advantages if nanoencapsulation technology in enabling OSD formulations that overcome low oral bioavailability due to biological barriers such as hepatic metabolism through cytochromes or high affinity with permeability glycoprotein (P-gp), a transmembrane transporter present in the intestine capable of pushing drugs back into the lumen, thus decreasing their absorption.
This is a significant issue for the pharmaceutical industry with an estimated 60% of new available APIs rated as Class II and Class IV under the Biopharmaceutics Classification System (BCS) based on their low water solubility or biological barriers.
Nanoencapsulation can achieve specific properties that are key differentiating factors, with nanoparticles acting as shields for the active molecule, preventing unhelpful binding to confer enhanced permeability. Nanoencapsulation can be applied to new chemical entities but also to existing market-approved APIs through product lifecycle management that gives pharma companies opportunities to increase the rentability of active ingredients by developing “premium” drug product generation with enhanced therapeutic efficacy and minimised off-target side effects.
NanoMicS Platform at a Glance
While widely used for injectables, nanoparticles remain poorly explored for oral administration.
To answer market needs, Skyepharma, which specialises in oral solid dosage forms and high-pressure homogenisation (HPH) technology, has created NanoMicS as a nanoformulation platform, focused on the development of innovative nanoparticle-containing tablets, capsules, or micropellets.
Skyepharma has already developed an HPH-based process capable of producing nano-sized particles of APIs named IDD-Dissocubes™. This top-down technology, resulting in nanocrystals for API, is currently being marketed as Triglide® for the treatment of for lipidic disorders with dramatically reduced food effect and administered dosage of fenofibrate, compared with traditional oral dosage forms.
Reinforcing the pipeline of bioavailability-improving solutions with nanoparticles manufactured through HPH is completely in line with Skyepharma’s strategy to bring high-value oral solutions to customers and ultimately patients.
The article explains how NanoMicS was developed in partnership with LAGEPP, which is highly experienced in nanoparticles and oral formulation processes. The platform started with the development of a generic nanoformulation to improve the solubility of lipophilic drugs, but there are plans to further expand the portfolio of nanoformulations to address all types of molecules, including temperature-sensitive APIs. All formulations are developed with “green” processes, avoiding the use of volatile organic solvents detrimental to the environment.
Recent Scientific Advances
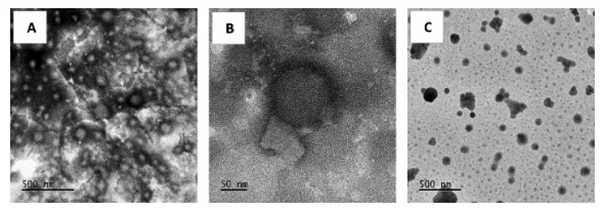
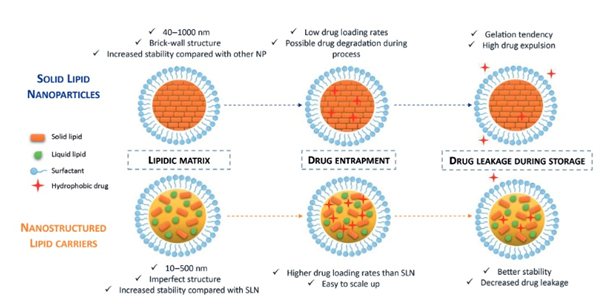
The article also discusses promising first results generated by researchers from LAGEPP on the nanoencapsulation of BCS Class II or IV APIs in solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs). This innovative strategy, improving the API’s solubility in digestive fluids and giving better control of its release and metabolism issues, also has the advantage of being biocompatible and biodegradable. Both SLNs and NLCs contain solid lipid, liquid lipid (Maisine® or Capryol® 90) and surfactant but differ in the whole structure, SLNs presenting a brick-wall structure while NLCs feature a hybrid structure.
The manufacturing of nanoproducts requires the control of nanomaterial properties such as size, shape, charge, composition, physicochemical properties and drug-release kinetics.
As a proof of concept study, a selected model drug spironolactone (SPI), a BCS class II API, was processed into a formulation of lipid nanoparticles developed according to a previous study carried out at LAGEPP using a nanosuspension composed of lipidic and aqueous phases heated separately and homogenised by high shear agitation. This pre-emulsion was inserted in the Microfluidizer® LM20 and pumped through the system, with high shear forces applied to the emulsion.
Both SLNs’ and NLCs’ blank formulas provided satisfactory particle size characteristics, with a mean diameter below 200 nm and polydispersity index (PDI) below 0.2nm.
The proof-of-concept confirmed that HPH can be used to manufacture lipid-based nanoparticles with mean diameters lower than 200 nm. While encapsulating the model SPI drug, the addition of a liquid lipid did not have a significant impact on the particle size but significantly improved the entrapment efficiency of the API, especially with Maisine. In vitro investigations are underway to study the release profile of the API as well as the improvement of its cellular permeability when encapsulated in lipid nanoparticles.
Industrial Scale-Up and QbD Approach
As a contract development and manufacturing organisation (CDMO) and center of excellence in oral solid dosage form development and industrialisation, Skyepharma is highly experienced in quality-by-design (QbD) and process industrialisation, which are two strong assets for the development of the NanoMicS platform.
For nanoparticle-containing tablets, attributes such as nanoparticle size, API encapsulation efficiency, polydispersity index and drug-release kinetics come on top of critical quality attributes that are standardly evaluated for tablets (hardness, friability, mean mass and disintegration). To increase manufacturing robustness, the process analytical tool approach should be considered at the industrialisation phase to allow real-time product analysis and continuous feedback on manufacturing.
“Defining clearly the attributes of raw materials, their behaviour throughout the process and their quality impact on the final product is also a prerequisite to the successful development of nanoparticles. In the case of solid lipid excipient, for example, changes in surface charge or morphology can alter the therapeutic properties of the API, and its characterisation is as important as the one of the API. Considering those industrial challenges early in development is a key element to reduce drug product development timelines and costs,” the article observes.
High solubility with lower environmental impact
“Skyepharma has made the strategic choice to establish itself as a pioneer in the administration of oral nanoparticles,” the authors conclude.
“Co-developed with LAGEPP, the NanoMicS platform aims to offer biocompatible delivery system solutions for BCS class II and IV molecules and reduce the number of leads of high therapeutic potential that are given up due to poor solubility issues. The Microfluidizer® technology appears as an eco-friendly solution to enhance oral bioavailability, broadening the spectrum of active molecules that could reach the market in oncology, immunology or infectiology and making patients’ daily lives easier,” they report.
About the authors
- Oksana Lemasson, PharmD, obtained a double degree in pharmacy and a master’s degree in industrial cosmetology, with a specialisation in formulation at the University of Lyon (France). Following her studies, she taught galenics for a year at university to pharmacy students. Wishing to develop her expertise in formulation, she is currently a PhD student at Claude Bernard University Lyon’s Laboratory of Automatic Control, Chemical and Pharmaceutical Engineering (LAGEPP), in partnership with Skyepharma, and works on the development of lipid-based nanoparticles, from proof of concept to manufacturing process optimisation.
- Sandrine Bourgeois is Associate Professor in Pharmaceutical Technology at the School of Pharmacy of the Claude Bernard University Lyon 1 (France). After graduating in pharmacy, she obtained a PhD in pharmaceutical technology at Paris-Saclay University (France).She conducts research at the Laboratory of Automatic Control, Chemical and Pharmaceutical Engineering (LAGEPP) on the development and characterisation of new drug delivery systems for oral and mucosal administration. Dr. Bourgeois is the author of 28 publications, three patents and has supervised 10 PhD students.
- Vanessa Bourgeaux brings 15 years of experience in research and development, and pharmaceutical development. She started her career at Erytech Pharma (Lyon, France), where she held the position of global project leader and actively contributed to the development of erythrocytes as drug carriers for oncology and sickle cell disease. In 2020, Dr. Bourgeaux joined Skyepharma. As Project Manager in the New Product Introduction department, her mission is oriented towards innovation and collaborative partnerships. Dr. Bourgeaux graduated in organic chemistry and holds a PhD in biochemistry and cellular biology. She is the author of 13 publications and 12 patents.
About Skyepharma Production
Skyepharma is an independent French pharmaceutical CDMO, 100% owned by its management team and Bpifrance. Skyepharma is an expert CDMO specialised in the formulation, development and manufacturing of complex oral solid forms (OSD), with a specific expertise and proprietary technologies on modified release products.
Skyepharma is based in Saint-Quentin-Fallavier, France. The current factory, dedicated to its OSD activity, occupies 22,000m2, on a 60,000m2 piece of land. Skyepharma has decided to allocate a portion of the available land (more than 20,000m2) to establish its SkyeHub Bioproduction, an innovative model designed to offer clinical and commercial production capacities to biotech companies. This SkyeHub model includes the construction of dedicated buildings, with specifically designed surfaces and premises, together with transverse support services such as quality, maintenance, batch release, etc.
Learn more at: www.skyepharma.fr
Resources
Click on Nanoencapsulation: a new era for oral solid dosage forms to access the full article.
Click on Triglide™ for example of Microfluidizer® drug application.