By Pharmatrans SANAQ…
Pharmatrans introduces SANAQ® SL 004 co-processed lactose/starch excipient
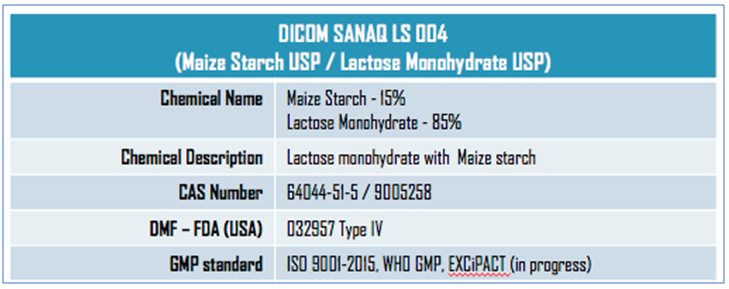
Pharmatrans Sanaq now offers SANAQ® SL 004 as an ultra-uniform and highly compressible lactose monohydrate and starch co-processed excipient for tablet fillings.
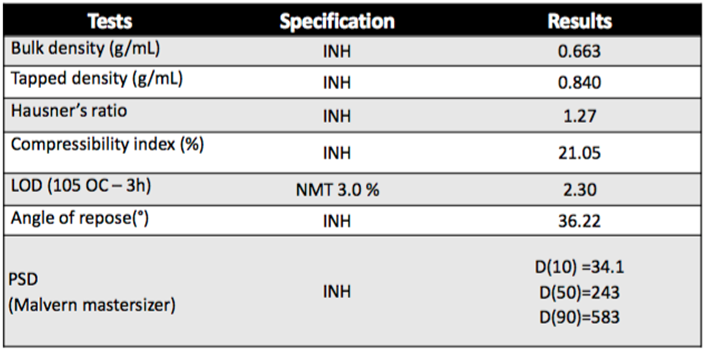
DICOM SANAQ® SL 004 has an average particle size distribution (PSD) of 300 to 550 microns and is designed for direct compression (DC) applications, aiding tablet hardness, faster disintegration, and superior flowability.
Superior flow and disintegration properties
SANAQ SL 004® is composed of lactose monohydrate (85%) and maize starch (15%) and exhibits excellent and free powder flowability compared with physical mixtures in the same ratio of components that show very poor flow properties.
As a further benefits offered, tablet disintegration of the tablets is independent from tablet hardness and lubricant level.
Resources
Click on Pharmatrans SANAQ Lactose/Starch SL 004 for further details.
Click on Pharmatrans SANAQ co-processed Lactose/Starch SL 004 for PDF download.