By Pharmatrans SANAQ…
Pharmatrans introduces DICOM SANAQ SP204 proprietary excipient with alkaline agent
Pharmatrans Sanaq has now added DICOM SP 204 tailored to help formulation development of moisture sensitive APIs and to stabilize formulations where alkaline conditions need to be controlled to its DICOM SANAQ® range of high functionality co-processed excipients.
SANAQ® SP 204 is a co-processed excipient optimized for direct compression and contains an alkaline agent that makes it especially suitable for acid sensitive APIs, along with diluent, desiccant, and disintegrant.
DICOM-DC® SP204 Features
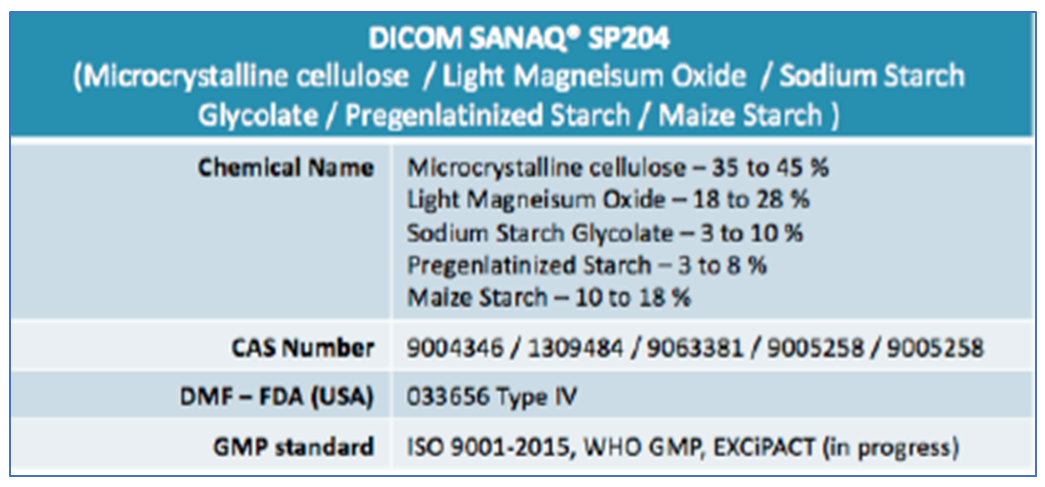
SANAQ® SP204 is a proprietary co-processed excipient containing Microcrystalline Cellulose, Light Magnesium Oxide, Sodium Starch glycolate, pre-gelatinized starch and Maize starch. It incorporates a large surface area of Light Magnesium Oxide to provide enhanced protection and stability to acid labile active pharmaceutical ingredients from a 360o alkaline environment.
The large Magnesium Oxide surface area in SANAQ® SP204 creates a 360° alkaline microenvironment at pH 9.5-11.0. This is essential in formulations involving acid labile drugs (e.g. Pantoprazole) to provide enhanced protection to the active ingredient.
SANAQ® SP204 also exhibits high bulk density making it suitable for formulation of high weight tablets.
SANAQ® SP204 Benefits
SANAQ® SP204 engineered to overcome a series of pre-existing Direct Compression challenges in incorporating Light Magnesium Oxide into a co-processed excipient, including the ‘fluffiness’ that made it difficult to incorporate directly into a tablet formulation, tendency to segregate, leading weight variations and unstable formulations, and inconsistent granular mix that could cause improper die filling and damage to tooling.
SANAQ® SP204 therefore offers a series of defined user benefits that include:
- Uniform alkaline protection from all sides
- Direct compression at high speed with uniform weight and quality attributes
- Enhanced stability for acid labile APIs
- Higher product quality in being able to form robust tablets with reduced friability
- Reduced tooling wear and damage through indirect contact and protection from other excipients in the co-processed mix
- No special coating requited for tooling
- High bulk density allows formulation for high tablet weight using smaller die or high fill weight in smaller capsules.
Resources
Click on Pharmatrans Sanaq DC SP204 for more information.
Click on DICOM SANAQ portfolio for a summary of available blends.
Click on DICOM SANAQ® solutions for direct compression formulations for full specification.