By Phage Consultants
Phage testing and therapy from Phage Consultants
Phage Consultants (PC) provides some of the world’s most expert bacteriophage consultancy services that can be applied to the fields of phage testing and phage therapy against bacterial infections.
Interest in phage therapy is rising in life sciences as the prospects of antibiotic resistance and over-use become mounting concerns. Accordingly, Phage Consultants offers this short guide to the evolving role of bacteriophages in medicine.
Introduction to Phage Therapy
Phage therapy (PT) is not a new science but was first developed more than a century ago during World War 1, when French-Canadian microbiologist Félix d’Hérelle, working at the Pasteur Institute in Paris, observed the case of a dysentery patient cured by administering a ‘phage cocktail’ to eliminate the bacterial infection.
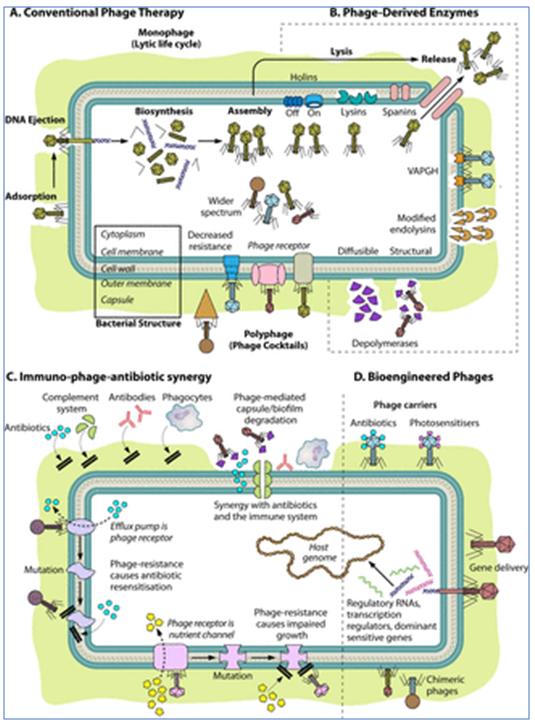
Essentially, phage therapy is the use of viruses to attack bacteria. Application of bacteriophages in this way carries a key advantage in that they only attack bacteria (the name means “eater of bacteria”) and cause no harm to flora and fauna.
While bacteriophages might look like alien invaders, they are an organic part of life on Earth, prevalent in soil, sewage and water and are nature’s way of keeping control of bacterial growth.
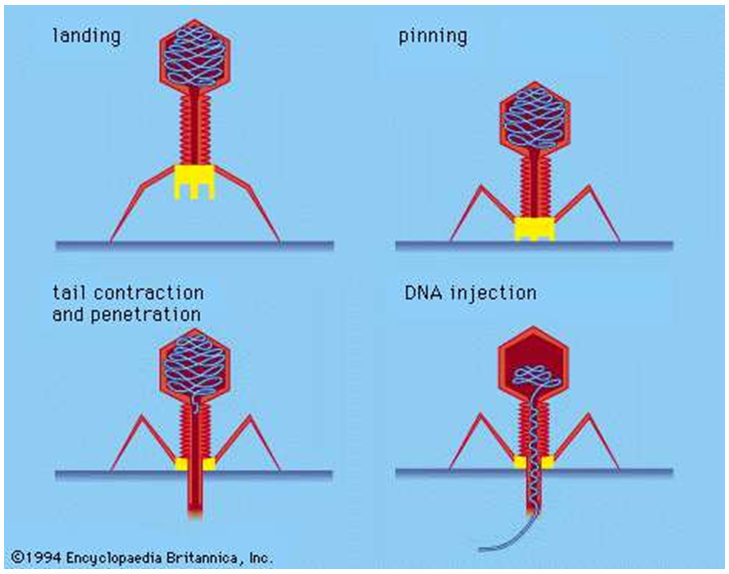
Bacteriophages kill bacteria by lysis, first binding themselves to the target bacterium and injecting their DNA or RNA into the cell. This causes copies of the phage to reproduce inside the cell, making as many as a thousand copies in each bacterium, which eventually bursts, releasing the new bacteriophages. Once bacteria are lysed, they are dead and cannot multiply. As with viruses in general, phages can remain inactive until suitable bacterial host ‘targets’ appear.
Relationships between phages and antibiotics
Antibiotics are man-made or naturally derived agents (e.g. penicillin) optimized to destroy bacteria and therefore effective in combating infections in humans and animals. However, they suffer from serious shortcomings.
One problem is that antibiotics tend to be less discriminate, attacking benign bacteria along with targeted harmful ones, leading to side effects such as digestive ailments and nausea. The second is that antibiotics are becoming less effective as bacteria develop resistance, leading to the rise of invulnerable ‘superbugs’.
Phages are now starting to be seen as a possible antibiotic alternative that suffers from neither of these disadvantages. They are far more selective in terms of bacteria they target, with each phage type related to a single bacterial class or group. In fact, ‘phage spot tests’ have been established as an effective method for identification of bacterial infections by observing which types of phage clear spots on bacterial culture lawns.
Finally, it is presumed that as ‘predators’ of the microbiome, phages have a natural and organic ability to evolve along with bacteria. Research on this aspect continues and it is not yet clear whether using phages in therapy might lead bacteria to develop ‘phage resistance’.
Phage therapy advantages
A 2011 research review, Pros and cons of phage therapy, by Catherine Loc-Carrillo and Stephen T Abedon, listed principal advantages and disadvantages of phage therapy. On the plus side, Loc-Carillo and Abedon found that:
- Phages can be effective against antibiotic-resistant bacteria;
- Phages can work as stand-alone therapies or in combination with drugs;
- Phages multiply organically, reducing dosage requirements;
- They have minimal side-effects against benign bacteria in humans;
- Phages are not toxic to humans, animals, plants, or environment.
Phage therapy disadvantages
On the minus side, the review suggested that phage therapy needs more research to find out how well it works. For example, it remains to be established that phages cannot harm people or animals in ways unrelated to direct toxicity.
Other identified disadvantages of phage therapy included:
- Difficulty in preparing phage-based treatments;
- Uncertainty over dosages or timescales;
- Challenges in identifying and isolating the most effective phage type;
- Uncertainty over phage effects on human immune system;
- Limited pool of identified phage types;
- Potential for bacteria to become resistant to specific phage therapies.
Current therapeutic use of phages
While neither the North American nor European regulatory bodies have yet approved phage therapy in human medicine, research of the best ways to apply bacteriophage technology in animals and people continues.
The prime focus of this research is in addressing infections that don’t respond to antibiotics, such as the ‘hospital killer’, Staphylococcus MRSA. There have also been anecdotal ‘freelance’ successes, such as the well-documented case from San Diego, California, of doctors using bacteriophages to cure a 68-year-old man, whose Acinetobacter baumannii infection had resisted all antibiotics.
Phage food additives and cleaning agents
Meanwhile, in the food industry phage therapy is becoming widespread with the U.S. Food and Drug Administration (FDA) approval of some phage mixtures to help stop bacteria from growing in foods. Phages are being added to some processed foods to combat food poisoning threats, such as Salmonella, Listeria, E. coli, Mycobacterium tuberculosis, Campylobacter, and Pseudomonas.
Another potential use for phage therapy under study is the addition of bacteriophages to cleaning products to destroy surface bacteria in hospitals, restaurants, and other sterile environments.
In all these various fields of research and application of bacteriophages, Phage Consultants with its world-leading expertise and accumulated libraries of phage types is able to bring valuable help and advice to all customers interested in phage testing and therapy.
Resources
Click on Temperate phage infection to see animation illustrating mechanism of action.