By MEGGLE Excipients
MEGGLE InhaLac®140 and InhaLac®150 milled lactose monohydrates for DPI applications
MEGGLE’s newest milled grades of lactose monohydrate, InhaLac® 140 and InhaLac® 150, mark a significant extension of its milled inhalative lactose portfolio. As mid-sized milled carrier lactose, both are suitable for use in pulmonary and nasal drug delivery.
These two new products perfectly fit in MEGGLE’s inhalative lactose grades product portfolio, completing the InhaLac® product family with milled carrier lactose grades for dry powder inhalation (DPI) applications.
Design & Features
Developed at MEGGLE’s Wasserburg laboratories in Germany, InhaLac® 140 and InhaLac® 150 are both new inhalative lactose grades with specific particle size distributions characterized by the typical flow- and surface characteristic of milled lactose grades. This provides an additional tool for the formulator to tune and optimize the performance of the DPI product.
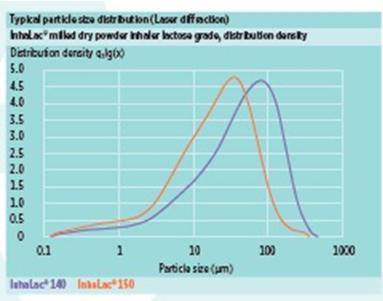
InhaLac® 140 is described by a mean particle size of approximately 50 μm, while InhaLac® 150 exhibits a narrow particle size distribution (PSD) with mean particle size of around 24 μm.
InhaLac® 140 features a PSD distributed thus:
- x10: 3-7 µm
- x50: 36-61 µm
- x90: 120-190 µm
Typical values for bulk density are 0.6g/mL and for tapped density 0.92 g/mL.
InhaLac® 150 features a PSD distributed thus:
- x10: 1-5 µm
- x50: 18-30 µm
- x90: 65-95 µm
Typical values for bulk density are 0.49g/mL and for tapped density 0.8 g/mL
Applications & benefits:
InhaLac® 140 and 150 are milled inhalative grade lactose monohydrates with distinct particle size distribution that extends MEGGLE`s product portfolio extension of inhalative lactose grades, closing the gap between coarse sieved and fine milled lactose grades for dry powder inhalation.
Particle size distribution differs from other products from MEGGLE`s product portfolio with specific particle size distributions characterized by the typical flow- and surface characteristic of milled lactose grades. This provides an additional tool for the formulator to tune and optimize the performance of the DPI product.
In summary, InhaLac® 140 and 150 offer distinct user benefits:
- Highly controlled and homogenous powder characteristics
- Highest microbial quality including low endotoxins
- High stability extends shelf life with retest only required after 24 months.
Quality & Certification
MEGGLE’s InhaLac® alpha-lactose monohydrate grades comply with the current harmonized Ph.Eur., USP-NF and JP monographs. InhaLac® 140 and 150 meet additional or stricter specification limits than those specified in USP-NF, along with Ph. Eur. and JP monographs to meet the special requirements for pulmonary drug delivery.
Additionally, MEGGLE is a member of IPEC (International Pharmaceutical Excipients Council).
The pharma-dedicated MEGGLE production facility in Wasserburg, Germany is certified according to DIN ISO 9001:2008 and has implemented GMP according to the Joint IPEC-PQG Good Manufacturing Practices Guide for Pharmaceutical Excipients and USP General Information Chapter <1078>.
MEGGLE invests considerably in raw material resource sustainability, production standards and efficiency and is actively engaged in environmental protection. Lactose meeting pharmaceutical standards is our first priority.
Specifications and regulatory documents can be downloaded from www.meggle-pharma.com.
Summary
InhaLac® 140 and InhaLac® 150 represent a significant extension of the MEGGLE portfolio of inhalative lactose grades, allowing milled DPI lactoses to be applied as carrier excipient to dry powder inhalation (DPI) formulations for which only sieved carrier lactoses were previously available.
Their distinctive surface characteristics and PSD patterns provide new possibilities for dry powder inhalation formulation teams.
MEGGLE can provide samples of the new powders to developers and researchers interested in experimenting using them in new formulations.
Resources
Click on InhaLac® portfolio for full product information.