By Insife
HALOPV Hosted on Insife.cloud: End-to-End Pharmacovigilance, Data Security, and Sustainability
Insife’s end-to-end global safety database, HALOPV, is a game-changing pharmacovigilance platform that provides organizations with the efficiency and automation needed for state-of-the-art PV process management.
When hosted on the innovative cloud-based IT platform Insife.cloud, HALOPV also benefits from added layers of security and integration with other cloud-based apps.
HALOPV: Pharmacovigilance oversight and advanced automation
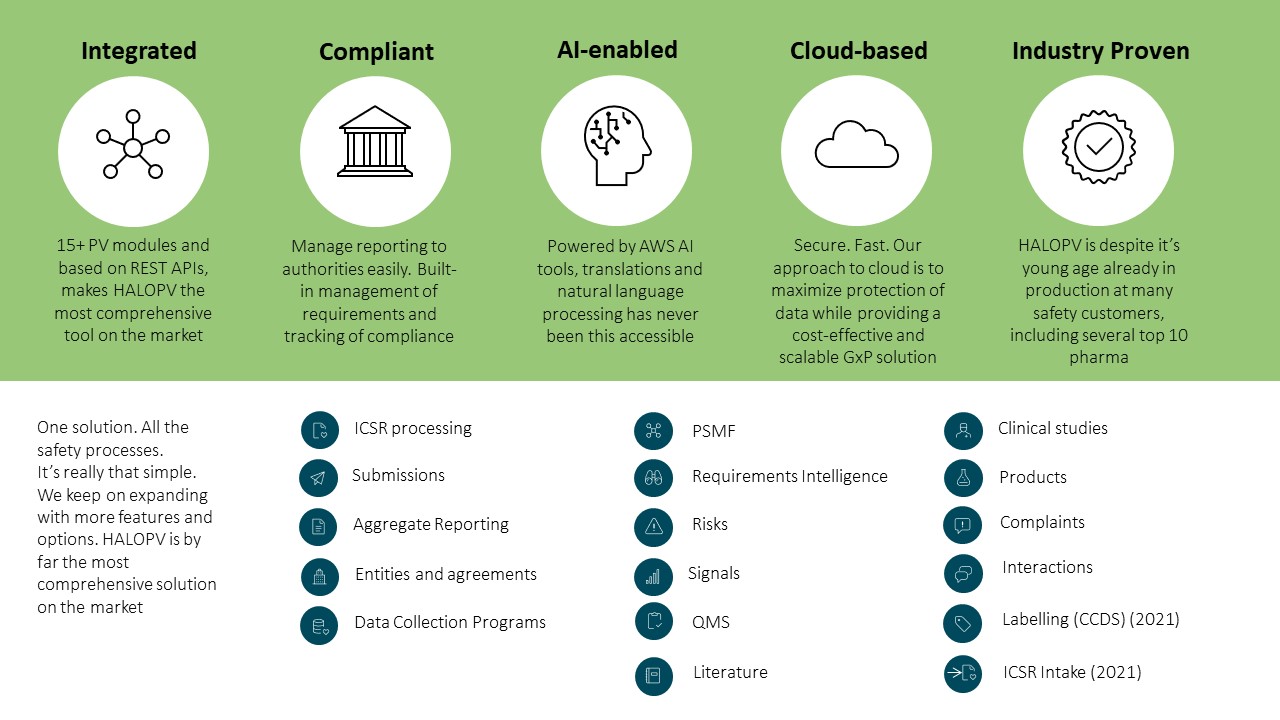
HALOPV is a full global safety database that features an efficient user interface as well as an advanced toolbox for automation as a “halo” around drug safety assurance for pharma and biotech companies and agencies. HALOPV provides comprehensive workflows and tools, such as artificial intelligence / machine learning, to ensure next generation drug safety automation.
HALOPV modules address every component of drug safety processing, including product safety master files (PSMFs), safety data exchange agreements (SDEAs), individual case safety report (ICSR), distribution and submission tracking, aggregate reporting, signals, risk management, literature, regulatory submissions, complaints, quality control, and even device vigilance (see Figure 1).
Meeting the global compliance challenge
HALOPV was developed in response to the challenges that pharma companies experience in maintaining fully compliant drug safety processes and documentation across global networks and markets. The application allows drug companies to orchestrate all their global pharmacovigilance activities through a single platform that automates tasks and speeds data flows and communications.
Traditionally, pharma companies have constructed their global PV response around a safety database with a number of additional stand-alone tools used to handle secure affiliate and partner reporting, compliance management, literature monitoring, regulatory intelligence, and risk management.
However, full automation has become increasingly important to meet the challenges of ever-increasing complexities in managing global operations.
Seamless integration
HALOPV features workflow-driven process support for global activities across pharma HQs, affiliates, partners, CROs, regulatory agencies. etc., with built-in compliance monitoring and management, including suggesting corrective and protective actions (CAPAs).
Built-in automation orchestration allows HALOPV to seamlessly incorporate rules-based as well as AI-based processing, e.g. used to manage unstructured sources with natural language processing (NLP) and reverse completion of language sections with natural language generation (NLG).
Advantages include full integration with Office 365 SharePoint or OneDrive environments, allowing users to generate Word, Excel, or PowerPoint documents etc. directly from HALOPV records in workflow and share with multiple reviewers.
Out-of-the-box integrations are available for Argus Safety database from Oracle™ and ARISg from Aris Global, for best-in-class data interoperability, as well as for analytics tools such as PowerBI from Microsoft or QlikSense from Qlik.
Mobile access and cloud hosting
HALOPV’s functionality extends to a mobile-friendly representation, supporting ready global access, anyhow, anywhere. Meanwhile data security is enhanced by cloud hosting, placing HALOPV on Insife.cloud to provide a total one-stop-shop for pharmacovigilance drug safety and GxP compliance solutions. This allows companies to implement drug safety processes and regulatory submissions faster, more cost-effectively, and in smarter ways.
The Insife.cloud platform provides access to an integrated suite of technologies and applications from a range of partners that also include the fully-featured Argus Safety database for fully integrated regulatory compliance tracking, aggregate reporting, and advanced workflow management. Insife.cloud also allows HALOPV to integrate with the Axway Gateway for regulatory submissions to authorities and Amazon Web Service (AWS) business tools such as AWS Translate neural machine language translation, AWS Comprehend NLP service that uses machine learning to find insights and relationships from unstructured data.
Safety, security and sustainability combined
Insife.cloud is fully GxP, GDPR and HIPAA compliant for ultimate global data security and optimal risk-protection, while offering system upgrades every year.
Cloud hosting provides a series of more general sustainability advantages that include superior power infrastructures with lower transmission losses, and reduced energy consumption (Lawrence Berkeley National Laboratory estimates cloud hosting reduces total energy consumption of business software applications by up to 87%). These benefits, along with higher utilization rates, faster upgrade and more frequent equipment refresh, also reduce total cost of ownership (TCO).
Sustainability and climate impact are also reduced because cloud providers tend to use cleaner power sources than average corporate data centers. For example, AWS uses a 28% less carbon-intense power mix. Insife is working towards ISO 14001 certification, making tangible its dedication to quality and sustainability.
Resources
Click on HALOPV for further information.
Click on Insife.cloud for further details.