By Insife
Insife HALOPV: The 21st Century Solution to Safety Reporting Challenges
Pharmaceutical companies face complex challenges in implementing efficient and responsive pharmacovigilance (PV) safety reporting. These challenges include (but are not limited to) the rise in caseloads, stricter regulatory expectations, trying to tie together multiple legacy IT systems, and finding the most efficient way to leverage AI and machine learning.
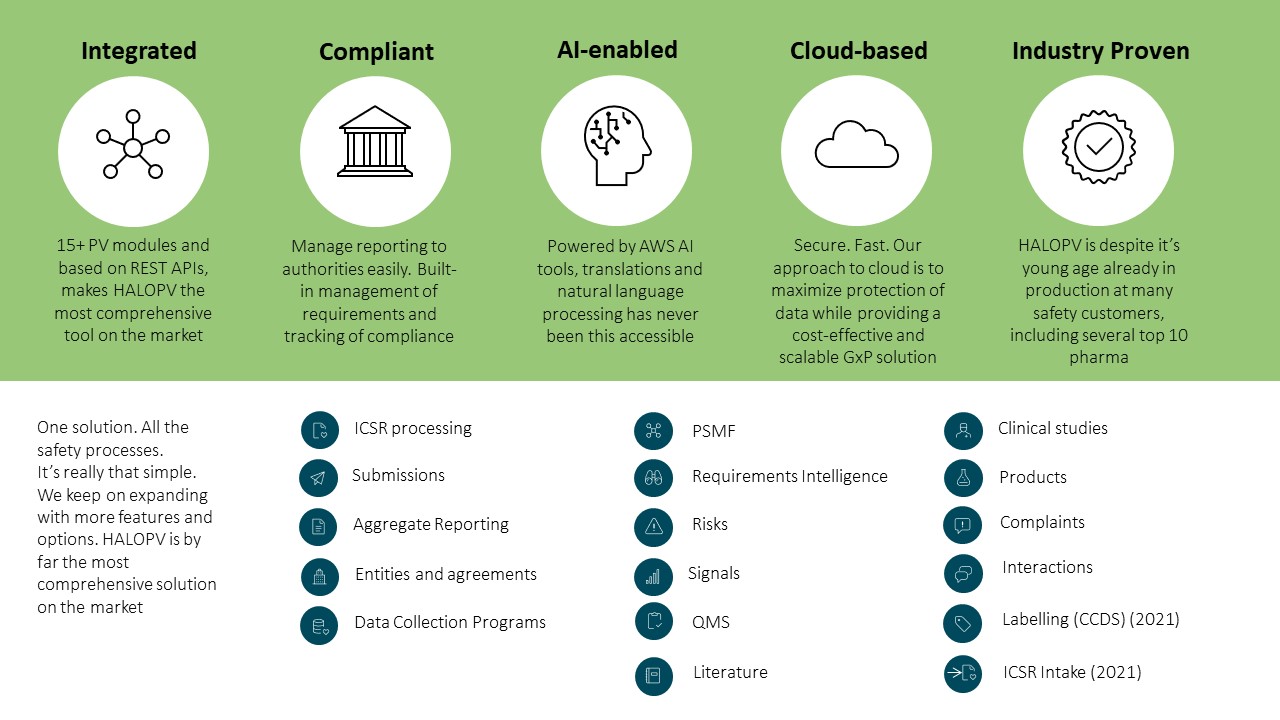
Insife’s HALOPV platform is a 21st century response to these challenges that re-imagines and revolutionizes drug safety automation, providing a full global PV pharmacovigilance safety database that features an efficient user interface as well as an advanced toolbox for automation as a “halo” around drug safety assurance for pharma and biotech companies along with regulatory agencies.
HALOPV functionality
The HALOPV system allows drug companies to orchestrate their global pharmacovigilance activities through a single platform that automates tasks and speeds data flows and communications.
HALOPV modules address every component of drug safety reporting, including pharmacovigilance system master files (PSMFs), safety data exchange agreements (SDEAs), individual case safety reports (ICSRs), aggregate safety reporting, risk management, regulatory submissions and complaints.
It provides comprehensive workflows and tools, such as artificial intelligence / machine learning, to ensure next generation drug safety automation that provides organizations with the efficiency and automation needed for state-of-the-art PV process management.
Meeting the safety reporting challenges
HALOPV was developed in response to the challenges that pharma companies experience in bridging between diverse safety databases and in maintaining fully compliant drug safety processes and documentation across global networks and markets.
It recognizes that one-third of total PV effort is expended on safety surveillance – signal detection, risk management and development/submission of aggregate safety reports.
Traditionally, pharma companies have constructed their global PV response around a pharmacovigilance safety database with a number of additional stand-alone tools used to handle secure affiliate and partner PV reporting, compliance management, literature monitoring, regulatory intelligence and risk management. The “cohesiveness” of surveillance processes is to a large degree reliant on manual ‘to-dos’, project plans and long trails of multiple emails and spreadsheets.
RSI and E2B tracking
Keeping reference safety information (RSI) updated and the link to labelling processes is difficult. Technology was required to make end-to-end RSI changes fully transparent and deliver needed metric insights to update, for example, the pharmacovigilance system master file (PSMF), without having to wait for a manual data collection at the end of every month.
While ICH compliant efficacy E2B(R3) submission tracking is comparatively easy, it is much harder with paper/pdf-based aggregate safety reports that are still used worldwide. Often, companies collect spreadsheets from each affiliate to collate a global overview, at a tremendous cost. Technology can be applied to track the status of local submissions, while also distributing CIOMS forms in an encrypted fashion, as well as aiding investigators and ethics committees requiring SUSARs and/or line listings.
Safety source management
One of the biggest hurdles of PV over the past five years has been the increasing focus on patient support programs and market research programs – failure to identify and start PV reporting safety information from these valuable sources can be detrimental.
Technology can provide a lean approach to registration, data collection and also local training of staff. Social media, clinical, reception calls etc. are all sources and a good system design also captures the information that does not qualify for getting imported into the safety database.
Integrated safety database and safety reporting
Full integration with Office 365 SharePoint or OneDrive environments allows users to generate documents directly from HALOPV records in workflow and share with multiple reviewers.
Further out-of-the-box integrations with Argus Safety database from Oracle™ and ARISg from Aris Global, along with analytics tools such as PowerBI from Microsoft or QlikSense from Qlik are also available for best-in-class data interoperability.
HALOPV’s functionality extends to a mobile-friendly representation, supporting ready global access, anyhow, anywhere. Meanwhile data security is enhanced by cloud hosting, placing HALOPV on Insife.cloud to provide a total one-stop-shop for pharmacovigilance drug safety and GxP compliance solutions. This allows companies to implement drug safety processes and regulatory submissions faster, more cost-effectively, and in smarter ways.
HALOPV: Pharmacovigilance oversight
HALOPV Version 3.0 introduces a new user interface based on modern design principles and full safety database capabilities to achieve compliance with FDA, EMA and other global regulators.
It now offers HALOPV Data mart for advanced querying and full Safety Database capabilities that can operate either in standalone mode or in integration with Argus, ArisG, etc. Insife pledges that HALOPV will remain fully open to integration with any vendor.
Its aggregate safety reporting features include:
- Managed schedules and workflow progress for aggregate safety reports
- Auto-generated report documents, formatted using Word templates
- Powerful workflow engine that allows locally generated various types of aggregate reporting from clinical to post-market while maintaining global oversight, review, approval, and management
- Built-in charts including Gantt overview of activities. Also monitor timeliness across all deliverables
- Link to Requirements Intelligence to ensure that aggregate safety reports are timely against regulations
Automated safety reporting
Full automation has become increasingly important to meet the challenges of ever-increasing complexities in managing global operations.
Orchestrating a strong process around intake of customer complaints and safety information, leading information through the right process steps, and capturing data efficiently, are formidable challenges. HALOPV provides a safety system that supports all aspects and makes it easy to prove compliance.
Built-in automation orchestration allows HALOPV seamlessly to incorporate rules-based as well as AI-based processing to manage unstructured sources with natural language processing (NLP) and natural language generation (NLG).
The Multivigilance approach
To overcome the challenges of global safety databases that are monolithic, outdated, and overly rigid, HALOPV provides possibilities to leverage creative automation by providing an adaptable, modular and therefore scalable model that meets the increasing need for a holistic ‘Multivigilance’ that goes beyond drug safety to integrate devices and data as well, opening up potential to apply IT automation, machine learning and AI across multiple channels and platforms.
The modular HALOPV approach also provides key advantages in that it is easier to keep pace with the ever-increasing rise in caseloads, currently increasing around 15 to 20 percent year on year, as well as the pace of evolving regulations, with their emphasis on standardizing and securing data.
This is fit-for-purpose PV Technology, supported by constant surveys of the technology landscape to understand current IT systems, level of integration, and identify areas for PV process automation.
HALOPV delivers a technology blueprint to achieve a compliant, high-performing and effective PV safety system.
Resources
Click on HALOPV for further information.
Click on Insife.cloud for further details.