By Erdmann Solutions…
Erdmann Solutions collaboration with Boreasa opens up new Respiratory Device markets
Neuhausen, Switzerland:- Award-winning design and engineering agency Erdmann Solutions AG (Erdmann) has helped to advance a revolution in respiratory therapy by playing a pivotal role in bringing innovative low cost powered medical devices to market.
Erdmann worked in three way collaboration with US-based Boreasa and contract manufacturing organisations to adapt the company’s industry-leading air blower technology to the need of healthcare markets.
Meeting Respiratory needs
In particular, Erdmann’s expertise in Early Stage Development was crucial in development of a range of Boreasa respiratory assistance medical devices suitable for individual and home care, meeting a global need underlined by the Covid-19 pandemic.
The Boreasa technology provides high power density and medical efficiency combined with the affordability that opens up a range of new home-care and other applications.
A range of Boreasa blowers applicable to Respiratory devices and ventilation systems was presented to the November 2022 Medica healthcare trade fair in Düsseldorf, generating enthusiastic industry reception and feedback.
Design partner
Erdmann Solutions, with its emphasis on Medical Design and Human Centered Design (HCD), has also become a long-term partner embedded in Boreasa’s independent R&D Team, contributing application, market and engineering knowhow that enables bespoke solutions to be brought faster to MedTech markets.
“This is a good example of Early Stage Development leading directly to successful medical production,” explained Erdmann Solutions co-founder Raimund Erdmann.
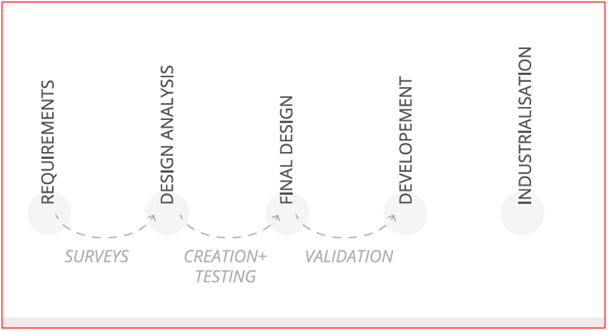
“Boreasa started out with an outline respiratory device concept. It was up to us to make that reality. That involved simulating and testing Boreasa’s initial concept against market realities and potential customers and user needs, leading to rapid prototyping to perfect different aspects of the design and manufacturing to gather further market feedback,” he continued.
Fellow Erdmann Solutions co-founder Raniero Pittini, who now leads the Boreasa team within the agency, described the early stage development process.
“At this stage, constructing single prototypes, it’s important to be able to use low-cost off the shelf components to keep overall development costs down and speed up. The prototypes lead us through a series of iterations to a final design that can be validated for functionality and regulatory compliance before being tech transferred to Boreasa for fabrication at their plant in China, using their own components,” Dr. Pittini explained.
The need for speed
“In this whole medical design process, speed is our biggest advantage, along with independence,” Raimund Erdmann reflected.
“We tend to attract early adaptors meaning that we need to test new markets with user feedback and use cases. That creates a need for speed because it isn’t until you can show something to medical doctors that works and put a price on it that you can fully find the markets for a new medical device,” he noted.
“And the independent mindset is important in working with a designer’s mind to challenge initial preconceptions and engineering skills plus marketing knowhow to identify new applications that might not have been obvious initially and can be patented,” he added.
Medical design with HCD methodology
Erdmann’s user-focused HCD methodology is an inversion of orthodox design approaches, placing the market first.
“The traditional way has been to ask the producer what is possible and then present that to the distributors in the market,” said Raimund Erdmann.
“We work in a different way, to find out what the hospital and customer need and then find the best ways to make that possible at a price he is happy to pay,” he explained.
In globalized markets, that includes taking into account different regional preferences and regimes that require the flexibility to provide different iterations and variants, as well as a system design approach in which patient safety is built in.
About Erdmann Solutions
Erdmann Solutions AG (Erdmann) offers award-winning medical design with human-centered Design and Engineering services that are particularly applicable to Life Sciences and Medical Precision Technology applications including pharmaceutical devices, patient monitoring systems, delivery robots, production systems and environmentally friendly packaging solutions.
The Erdmann Solutions Approach are built around Innovation, Human Centric Design underpinned by usability engineering and testing and Branding Expertise, bringing tightly knit and dynamic design teams to focus on innovation and real customer needs, with particular expertise in high-risk in technologies in Medical and Pharmaceutical Procedures.
Erdmann Design Solutions was founded in the Swiss canton of Aargau, close to Zurich, in 1978 by industrial designer Raimund Erdmann, who still leads the company with the managing partner Raniero Pittini. Since then, it has won more than 65 outstanding awards for its work with companies worldwide.
Further information at: https://erdmann.ch/
About Boreasa
Utah-based Boreasa has a name derived from the Ancient Greek God of Wind, now established as a brand for high performance miniature blowers, fans and drivers.
Boreasa’s mission is to design and manufacture high performance and highly reliable air moving products with an emphasis on quality and affordability that open up new solutions for consumer, medical, industrial, and military applications in global markets. Its products have been exported to the United States, Brazil, Argentina, Hungary, Israel, Egypt, India and other countries.
Boreasa combines the dynamism of Western design and engineering with low-cost Asian manufacturing. Its innovative blowers, motors and impellers are designed in California and Switzerland and manufactured in a state of the art ISO 9001:2015 certified plant in China.
Boreasa offers a wide range of standard line of products and also works with customers to develop customized products for specific applications. Its international R&D team with completely independent intellectual property rights ensures product quality to meet the most demanding regulatory standards.
Resources
Click on Boreasa at MEDICA Dusseldorf for further information.
Click on Experience Mapping for Human Centered Design to download PDF.