By enGenes Biotech…
enGenes secure significant funding for ECOnti E. coli Continuous Manufacturing Project
Vienna, Austria: – The Austrian Research Funding Agency (FFG) has awarded significant funding to recombinant proteins specialist CDMO enGenes Biotech GmbH (enGenes) to lead a consortium to advance continuous manufacturing based on Escherichia coli (E. coli).
The FFG has committed to providing total funding of 2.7 Million EUR for the ECOnti project, which has been estimated to have total costs of 3.6 Million EUR to reach the final process.
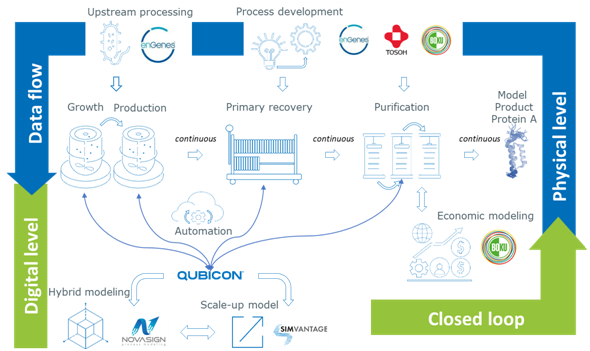
The project “ECOnti – Accelerated, low ecological footprint, manufacturing platform for continuous production of biotechnological products” aims to develop a fully integrated and automated continuous upstream and downstream process at up to 10 L batch size.
This small-scale development platform will enable the industry to develop continuous bioprocesses and implement model predictive control and the consortium will also perform economic modelling to showcase potential cost-savings of continuous manufacturing vs. batch-based manufacturing.
Initial life-cycle engineering (LCE) economic analysis has indicated substantial potential bioprocess operating cost advantages when compared with discontinuous batch processes.
Six member consortium
enGenes will contribute its patented -eXcite technology for continuous two-stage manufacturing, as well as its proven high-yield -eXpress expression platform, while the Vienna-based University of Natural Resources and Life Sciences (BOKU) will provide expertise in developing continuous primary recovery and continuous chromatography.
The remaining four consortium members are:
- Tosoh Bioscience: providing expertise in continuous DSP and its brand-new OCTAVE® Bio technology
- Qubicon AG: adapting its Qubicon process automation and control platform to the project
- Novasign: software and expertise for hybrid modelling
- SimVantage: software and expertise for CFD modelling.
ECOnti will form a quasi-autonomous body with dedicated project management and communications, including a website.
Overcoming CM barriers
So far, the biotech industry has been unable to implement continuous manufacturing (CM) using E. coli or other bacteria due to low process stability and consequently product yields.
The FFG-funded project aims to overcome these barriers and in addition develop a small-scale process that will establish model predictive control to dominate a fully integrated process at full scale.
enGenes recently established a method for continuous upstream processing (USP) of E. coli. The project will help to establish a valid proof-of-concept for fully integrated continuous bioprocessing by adding the expertise of its partners in digitalization, modelling, chromatography, etc.
Pioneering project
enGenes Biotech CEO Dr. Juergen Mairhofer commented:
“This is a unique consortium with complementary expertise. The problems addressed within ECOnti have not been tackled so far. The consortium will be the first worldwide to establish a fully integrated continuous bioprocess for Escherichia coli, thereby providing a blueprint for the cost-effective, ecological manufacturing of recombinant proteins or other biomolecules.”
Project leaders envision the technology developed within ECOnti will be key for biopharma, industrial biotech as well as the food and feed industry in offering lower manufacturing costs and a small footprint model, based on key advantages:
- Requires smaller production facilities
- Reduced water consumption
- Lower energy consumption and CO2 emissions
- End-to-end process control
- Fully digitized (digital twin by modelling) and automated
- Higher product yields and enhanced process stability based on proven enGenes proprietary -eXpress and -eXcite technology.
Leading role
Dr Mairhofer said the funding of ECOnti marked a further stage in the development of enGenes.
“Our success in raising non-dilutive funding for a project recognized as significant acknowledges the intellectual capital we have brought to the table,” he said.
“We are deeply proud to be leading such an impressive consortium of brilliant companies and scientific teams, together forming a research team with the power to solve relevant industry problems,” Dr Mairhofer added.
“We are all very clear in our ultimate mission: to provide the industry with access to biotech products with a lower ecological footprint at affordable prices,” he concluded.
About enGenes Biotech
enGenes Biotech GmbH (enGenes) is a contract research, development and manufacturing company that provides leading-edge technologies and production services focused on recombinant proteins in bacteria. The company’s mission is to provide cost-effective and scalable production of recombinant proteins at a fraction of the current cost, allied to a vision of developing a world-class portfolio of cutting-edge protein production technologies, relevant to a broad spectrum of application fields.
enGenes has developed advanced technologies to drive more cost-effective recombinant protein production processes, including its proprietary enGenes-eXpress™ E. coli platform that achieves outstanding yields of soluble and active recombinant protein. enGenes-eXpress™ has been successfully applied for the manufacturing of enzymes and biopharmaceutical products that failed to give economically feasible yields in a conventional expression host.
enGenes Biotech offers development and manufacturing services tailored to the needs of pharmaceutical and industrial biotech companies. The services include expression strain and vector development, fermentation process development and optimization, downstream process development, production of purified protein, technology transfer and scale-up support with technology out-licensing and co-development opportunities.
Resources
Click on enGenes presents -eXcite technology for further information.