By enGenes Biotech…
enGenes development and manufacturing support
enGenes Biotech offers development and manufacturing support services that are highly relevant to companies wishing to develop cost-effective manufacturing processes for recombinant proteins.
enGenes can assist clients to rapidly construct economic, scalable and efficient production processes based on its world-leading enGenes-eXpress technology for growth-decoupled protein production that provides powerful efficiency advantages for added value and commercial edge.
enGenes offers a modular system of customer services for gene expression and protein production in Escherichia coli .
Modular service platform
Its services are particularly useful for producing difficult-to-express proteins. The service platform is modular, being divided into a series of discrete steps that can be performed individually or in combination to cover all specific demands along the line from gene to protein.
The range of service options cover:
- Cloning / Vector optimization
- Generation of expression strain / Host cell engineering
- Feasibility studies for application of enGenes-eXpress technology
- Techno-economic analysis (TEA) of the planned production process
- Lab scale fermentations and bioprocess optimization
- Establishing of purification protocol and final purification
- Scale-up runs
enGenes project support plans also meet individual documentation requirements to facilitate easy tech transfer to the client’s own facilities and quality management system.
Biotech process development
Bioprocesses, harnessing the properties of cells and enzymes through tools such as recombinant DNA technology to develop novel products, presents a series of challenges, particularly in downstream processing and scale up to industrial viability.
Definitions, activities, and things to consider when developing an upstream or downstream biotech process, defined as the activities required to produce a biomolecule, be it a antibody fragment (FAb, scFv, VHH or any other antibody mimetic), recombinant protein, subunit vaccine, or any other product with a biological origin.
Bioprocess development is usually divided into upstream process (USP) development and DSP downstream development. Both activities must be supported by rigorous analytics, to ensure the product’s critical quality attributes (CQAs) are preserved through the whole process.
Early stage (preclinical) drug development simply requires a process adequate to meet laboratory quality needs. However, it also should be designed for an end goal of economic production at commercial quantities, being capable of easy scaleup through Phase I/II early clinical stages to full scale industrial scale. By the time late clinical Phase III is reached, the manufacturer will need robust USP and DSP biotech processes that deliver high yield and productivity while remaining cost-effective and reproducible. It is really advisable to “start with the end in mind” with regards to bioprocess development.
Market imperatives for manufacturing speed and economy translate into growing pressure to quickly create cost-effective process scale manufacturing. A combination of incremental gains in productivity can have sizeable impact on overall process economy.
Manufacture of a typical recombinant DNA-derived product will involve up to 20 steps between identifying basic source materials or tissue and final packaging, with the first half of the process concerned with genetic manipulation of the host organism. Cloning a gene from DNA into Escherichia coli (E.coli) will involve genetic engineering at laboratory scale to establish levels of expression available and the stability of the constructed strains.
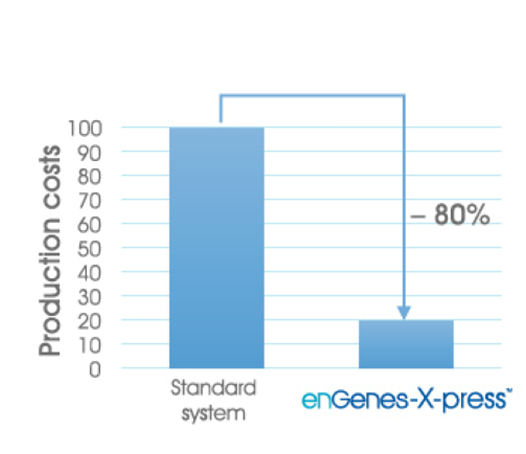
The fully developed culture must then be suitable for tech transfer to pilot scale bioreactor, where equipment design or configuration may affect process viability. Full process scale contract manufacturing also needs to consider downstream process (DSP) development and product recovery, which can be a major source of added cost and lost efficiency. For some recombinant-DNA-derived products, DSP biotech purification can account for 80 to 90% of the total processing cost.
-eXpress advantages
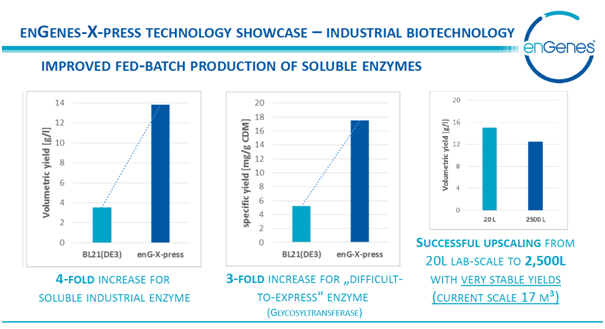
The unique enGenes-eXpress™ proprietary technology platform is leveraged from enGenes’ world-class experience in cutting-edge genetic engineering, combined with a broad expertise in bioprocess engineering, to produce a range of best-in-class recombinant protein expression systems.
The -eXpress technology fermentation platform is based on a genetically modified host cell based on BL21 (DE3) that allows introduction of standard expression vectors (with T7 promoter) with no need for cumbersome adaptations. The process can be implemented with standard equipment supporting easier protein expression scale-up and scale-up fermentation.
The platform meets the full range of development and manufacturing needs, including USP and DSP development for recombinant protein production, site-specific labelling of recombinant proteins using expansion of the genetic code and non-canonical amino acids, USP and downstream process development for plasmid DNA production, continuous process development, process optimization and scale-up and non-GMP production.
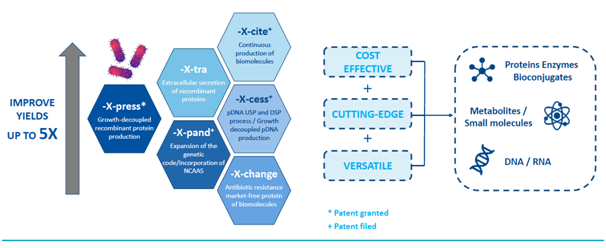
-eXpress is a platform technology that can be intelligently applied in many ways to produce proteins, enzymes, bioconjugates, metabolites, small molecules, and plasmid DNA (pDNA) or RNA. These related technologies include:
- -eX-tra for extracellular secretion of recombinant proteins
- -eX-pand for expansion of genetic code and incorporation of non-canonical amino acids (NCAAs)
- -eX-cite for continuous production of biomolecules
- -eX-cess for plasmid DNA (pDNA) upstream and downstream process development and growth-decoupled pDNA production
- -eX-change for antibiotic resistance market free protein of biomolecules.
Even in advance of the project research phase, enGenes is able to liaise with customer’s chosen CDMO or CMO manufacturing organisation to ensure that the developed scale-up process exactly fits with the capabilities of the CMO. It will ensure that the tech transfer process runs smoothly with upstream processing in biotechnology and downstream pre-developed and precise yield estimates for fed-batch processing so that client gets the best value for money. By starting with “the end in mind” enGenes can make sure that the economic goals of its clients are met and that the product becomes economically viable.
Resources
Click on enGenes production partnering for more information.