By enGenes Biotech…
enGenes Biotech’s continuous microbial protein expression and purification
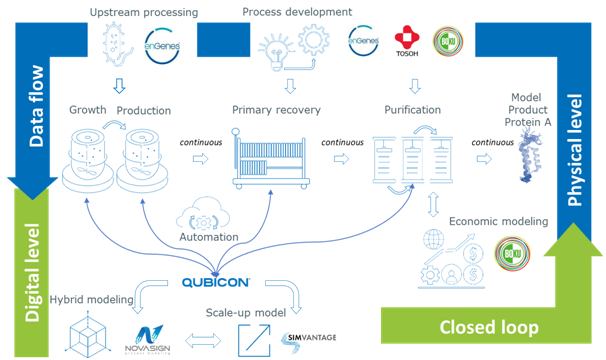
enGenes Biotech is implementing upgrades to its processes and equipment that will allow it to offer continuous recombinant protein purification and expression in Escherichia coli.
This significant upgrade marries enGenes’ patented -eXpress™ and -eXcite™ platforms for enhanced yield expression and continuous protein expression/purification and production of biomolecules in E.coli with new processes and technologies acquired as results of its lead participation in the Austrian federally funded ECOnti project.
As a result, enGenes is re-integrating all unit operations to accelerate its microbial protein production and protein purification.
Advanced MCC protein purification
One of the most significant of these is installation of the Tosoh Biotech Octave BIO from Tosoh Bioscience as one of the world’s most comprehensive and versatile multi-column chromatography (MCC) systems for intensified biologic processes that also unlocks resin and buffer savings, and eases scale-up to GMP-ready protein purification protocols.
The system features six independently driven pumps each assigned to a process fluid, with different pump sizes to cover a wide range of process scales. Built-in UV, conductivity, and pH detectors enable complete protein purification process monitoring, while onboard sample injector, mixer, and gradient capability enable process design experiments as well. A biocompatible multi-way valve block design allows for connection of up to eight LC columns to accommodate increasingly intensified upstream titers.
ECOnti protein purification protocols
enGenes is also able to incorporate other protein purification steps and processes learned as part of the ECOnti project to develop an accelerated, low ecological footprint, manufacturing platform for continuous production of biotechnological products.
This has progressed very rapidly towards its aim of developing a fully integrated and automated continuous upstream and downstream process at up to 10 L batch size, which is what enGenes will be able to offer by the end of 2024.
This will enhance the core enGenes offer of high yield expression of difficult-to-express proteins using a modular platform that can be divided into a series of discrete separation steps performed individually or in combination.
The range of service options covers all specific demands along the line from gene to protein, including:
- Cloning / Vector optimization
- Generation of expression strain / Host cell engineering
- Feasibility studies for application of -eXpress™ suite technologies
- Lab scale fermentations and bioprocess optimization
- Establishing of protein purification protocol and final purification
Protein purification and expression in -eXpress™
The unique enGenes-eXpress™ proprietary technology platform is leveraged from enGenes’ world-class experience in cutting-edge genetic engineering, combined with a broad expertise in bioprocess engineering, to produce a range of best-in-class recombinant protein expression and purification systems.
The fermentation platform is based on a genetically modified E. coli host cell that allows introduction of standard expression vectors (with T7 promoter) with no need for cumbersome adaptations. The process can be implemented with standard equipment supporting easier protein expression and fermentation scale-up.
The -eXpress™ core technology can be intelligently applied in many ways to produce proteins, enzymes, bioconjugates, metabolites, small molecules, and plasmid DNA (pDNA) or RNA in conjunction with related enGenes technologies that include:
- -eXtra™ for extracellular secretion of recombinant proteins
- -eXpand™ for expansion of genetic code and incorporation of NCAAS
- -eXcite™ for continuous production of biomolecules
- -eXcess™ for pDNA upstream and downstream process development and growth-decoupled pDNA production
- eXchange™ for antibiotic resistance marker-free recombinant protein and plasmid DNA production
Along with continuous process development, these platforms meet the full range of development and manufacturing needs, including USP and DSP development for recombinant protein production, site-specific labelling of recombinant proteins using expansion of the genetic code and non-canonical amino acids, USP and downstream process development for plasmid DNA production, process optimization and scale-up and non-GMP production.
Resources
Click on Cost-effective protein production to learn more.
Click on Octave® BIO Specification Sheet for detailed information.