By MEDELPHARM
CMAC Open Days to feature MEDELPHARM as part of Future Manufacturing Research
Beynost, (Lyon) France: – STYL’One compaction simulation instruments from MEDELPHARM were a prominent feature when the University of Strathclyde Centre for Continuous Manufacturing and Advanced Crystallization (CMAC) placed its current research projects on show at its recent Open Days.
CMAC’s series of three Open Days commenced on November 14 at University of Strathclyde which showcased its Future Manufacturing Hub’s development of the DataFactory solution in which MEDELPHARM is a collaborator.
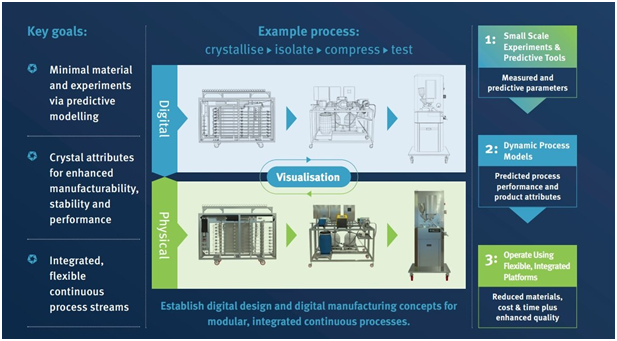
STYL’One inputs to QbDD
MEDELPHARM tablet research and development technology in the shape of its STYL’One Evo and compact Nano instruments are used by CMAC’s Future Manufacturing Research Hub scientists as part of their aim to revolutionize the development and supply of functional, high-value chemical and pharmaceutical products by delivering a rapid, digitally enabled pipeline to integrated continuous manufacturing processes.
One of the key parts of the Hub’s work is its development of a Quality by Digital Design (QbDD) approach to end-to-end processes. The QbDD workflow includes the concept of Digital Twins for design and operation of continuous processes, and the use of models to identify robust design spaces and inform control strategies that will be used to implement integrated continuous processes using the Hub’s DataFactory and MicroFactory platforms.
Digital Visualization
The team’s work has potential to revolutionize many aspects of pharma manufacturing, including API particle formation via crystallization, filtration, washing, and drying, along with drug product secondary processing including powder compaction and tableting.
The model-driven autonomous crystallization DataFactory research focuses on automating experiments based on data gathered from STYL’One instruments to deliver large, structured data sets for interrogation by image analysis and machine learning. These crystallization experiments will sweep through physicochemical phase space from molecule to solubility, kinetics, growth, agglomeration and fouling. The data collected will be used for research on image analysis, model development and machine learning. This will feed into work to establish a Crystallization Parameter Database.
Webinar presentation
A leading figure in developing the DataFactory is Dr. Daniel Markl, who will form part of the discussion panel at the CMAC Open Days. Dr. Markl will also present the QbDD-driven concept in a MEDELPHARM webinar, presenting a self-driving, high-throughput, data-intensive micro-scale development system as a tableting ‘DataFactory’ that can replace time consuming, costly and inflexible conventional methods for testing tablets.
He will show how system combines MEDELPHARM’s STYL’One compaction simulator with automated dosing and dedicated powder transportation units, near-infrared spectroscopy for evaluating powder blend homogeneity, and an automated testing system for measuring tablet properties.
The Self-driving tableting DataFactory webinar will take place from 1500 hrs CET (UTC +1) on December 5, 2023, with free registration available online (see Resources).
Using structured data
Dr. Markl will describe how data gathered from a STYL’One instrument can be automatically structured and fed into a database for the development of a hybrid system of models, including mechanistic and data-driven (AI) approaches, using raw material properties to predict critical powder blend characteristics such as flowability and tablet attributes, including tensile strength and porosity. The system will be able to automatically prepare and measure powder, produce and test single tablets explore advanced formulations and processes for manufacturing bilayer and multilayer tablets.
The webinar will further discuss the combination of hybrid modelling approaches with model-based optimization and the micro-scale tablet development system to reduce hands-on-lab time by more 80 per cent, with additional reductions in material use and waste.
About MEDELPHARM
Headquartered near Lyon, MEDELPHARM is a high-precision engineering company manufacturing R&D tablet presses and providing innovative solutions for powder analysis and formulation.
Since 1985, MEDELPHARM have been specialists in ‘Made by people for people’ tableting instruments that work with high-speed and precision but at the same time are very easy-to-use.
Since the end of 2022, it has been a member of the international KORSCH Group, enhancing its worldwide reach.
With its historical background in actual production, MEDELPHARM has a deep understanding of the R&D requirements that apply to tableting processes.
For formulation scientists and pilot plant developers around the world looking for assistance in powder characterization, solid dosage formulation or production trouble shooting, MEDELPHARM can offer a comprehensive portfolio of multi-functional tableting Instruments, R&D presses and high-speed compaction simulators as well as process equipment and extensive Science lab services in tableting, early research API characterization all the way to product development.
For more information, visit: www.medelpharm.com
Resources
Click on MEDELPHARM Self Driving Tableting Data Factory webinar for further details.