By Gerteis Maschinen…
Choosing a compactor for pharmaceutical applications
A roller compactor is pharmaceutical machinery used for tablet manufacturing, as well as powdered fillings for sachets and capsules. The purpose of these machines is to agglomerate the powders containing active ingredients and excipients to form uniformly sized granules. This entails subjecting the powder to precisely metered crushing and grinding forces between rotating rollers.
Choosing the right roller compactor is a matter of matching its features and engineering to target application and individual needs.
Pharma Roller Compactor applications
Traditionally, dry granulation in tablet manufacturing was performed by ’slugging’ – feeding the blended granules direct into a tablet press, to be crushed and compacted under pressure in a forming die. The main issues with slugging are that fine particles do not flow well into the die, leading to unacceptable variations in tablet form and cohesion. Consequently, roller compaction has largely replaced slugging in dry granulation.
The roller compaction process usually involves feeding various sized particles of active ingredient plus excipients like lactose or cellulose between two textured press rollers that then produce a compacted powder called ‘ribbons, ‘flakes’ or ‘briquettes’. These can then be further milled to a uniform and cohesive powder, ready for final tablet manufacturing.
Use of laboratory roller compactors in formulation development, helps to overcome challenges of inconsistent bulk properties and fine powder characteristics to adapt substances to tableting or fillings.
Use of a roller compactor allows for higher performance manufacturing of capsules and tablets. Roller compaction improves the bulk density of the processed powders significantly.
Primary considerations when purchasing a granulation pharma compactor are its total costs of ownership (TCO), ease of cleaning, suitability for high containment manufacturing and suitability for processing target substances and ingredients. The latter consideration will be impacted by roller design.
As with any pharmaceutical equipment, there are operational challenges when using a roller compactor as granulation equipment. These include overcoming loss of recompactability (with effects on tensile strength) and formation of non-compounded powder residues in dry granulation.
Gerteis® Roller Compaction Engineering
Gerteis Maschinen + Processengineering AG (Gerteis®) has become a world-respected name in pharmaceutical roller compaction, backed by more than three decades of constant development and innovation.
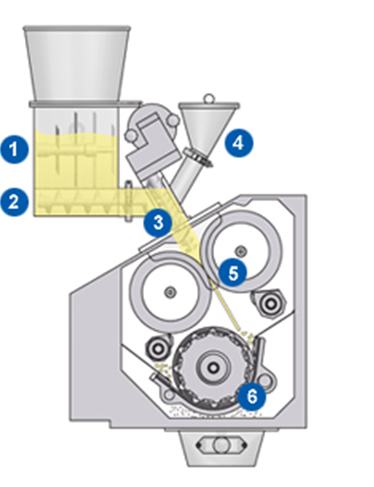
A GERTEIS® roller compactor comprises three principle sections or sub-assemblies.
- The feeding system, conveying uncompacted particles to the compaction unit and feeding the powder between the rolls. The feed hopper is the point of entry of all the materials used in the granulation pharma compactor process.
- The compaction unit, where granules are compacted between two counter rotating rolls, applying precisely defined force and a metered gap between the rollers to produce a ribbon of crushed particles.
- The size reduction unit in which the ribbons are milled to desired particle size.
Different roller compactors have different technical specifications that manufacturers need to consider before choosing a compactor for their particular application.
Many roller compactors on the market use an adjustable ‘fixed gap’ between the rollers. This system is robust but can draw in inconsistent amounts of powder between the rolls, with varying forces applied to the powder bed, causing fluctuations in the ribbon and granulate properties.
Gerteis® roller compaction units use a floating gap in which the distance between the rolls changes according to the amount of powder fed between them. This ensures the rollers apply a constant compaction force to ensure minimized fluctuations in granule properties.
Vertical ‘crushing’ force onto the granules the key parameter in the dry granulation compactor process, and this should be expressed in kilonewtons per centimeter (kN/cm) of roll width, rather than bars of pressure, which only represents the hydraulic pressure within the system, rather than the actual compaction force applied to the powder.
Roller speed is also important in the granulation pharma compactor process since it will determine the size of the granules formed.
To ensure that its roller compactors can control and measure all product quality relevant parameters (e.g. force, gap and roll speed), GERTEIS® has developed its Process Analytical Tool (PAT) in which all key compaction parameters are calibrated and validated. PAT also powers Gerteis® pharmaceutical machinery control systems to help manage batch-to -batch variations and ensure uniform granule properties during manufacturing.
Gerteis® roller compaction solutions
Gerteis® pharma roller compactors are specifically designed to meet dry granulation pharma compactor needs and also support ultra-high pharma containment requirements meeting very low occupational exposure (OEL).
Gerteis® offers five different pharma roller compactor solutions, tailored for a range of needs from proving laboratories to high containment roller compaction.
- MACRO-PACTOR®: The Gerteis® flagship pharma roller compactor solution, for scale-up from development to high speed production at up to 400kg/h and offering a very wide range of press force, roller speed and gap settings. Engineered around an open star rotor, closed pocket type granulator with integrated bulk-breaking and precrushing for optimal compaction results, MACRO-PACTOR incorporates interfaces for pneumatic or mechanical conveyor systems, feed and discharge for high containment compaction applications.
- MINI-PACTOR®: Gerteis® ’ compact laboratory roller compaction package designed and engineered to handle pilot projects and small scale production batches, also offering the low OEL levels, under 1 µg/m3, and sealed interfaces needed to support pharma containment compaction applications. Substances that have successfully been processed on MINI-PACTOR can be directly transferred to MACRO-PACTOR for high scale production without loss of quality.
- ULTRA-PACTOR®: a high-end pharma roller compactor engineered for ultra-high pharma containment applications, supporting OEL levels of under 0.1µg/m3, while still offering throughputs up to 400kg/h.
- POLYGRAN®: A pharma roller compactor oriented towards production and value for money, featuring simplified handling and controls for high accessibility. Ingeniously engineered powder feeding and parts assembly. This allows POLYGRAN to support medium production quantities with throughputs of up to 300kg/h.
- MINI-POLYGRAN®: an ultra-compact and highly mobile pharma roller compaction solution that strikes an ideal balance between performance and flexibility. Unique POLYGRAN engineering allows ultra-rapid maintenance and batch change, with production volumes of up to 45kg/h. The machine can be supplied with four-castor mobile trolley frame for flexible placement within the laboratory.