By SIRION Biotech…
SIRION LentiBOOST™ non-cytotoxic transduction enhancer for lentiviral vectors
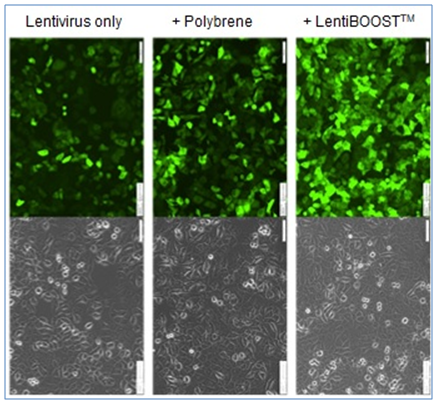
SIRION Biotech has developed LentiBOOST™ as a highly effective, non-cytotoxic transduction enhancer for lentiviral vectors in preclinical and clinical applications.
As a universally acting and receptor independent adjuvant, LentiBOOST™ can be applied to a wide range of clinically relevant cell types, making it a promising candidate to improve clinical transduction protocols for ex vivo gene therapies and CAR-T cell therapies.
With up to five-fold increases in transduction, LentiBOOST offers powerful benefits for biotech and pharma companies, drug developers, academic researchers seeking improved results in CAR-T clinical trials, cell therapy research and development studies.
LentiBOOST™ features
LentiBOOST™ is a highly effective, non-cytotoxic transduction enhancer for preclinical and clinical application of lentiviral vectors. As universally acting (receptor independent) adjuvant it can be applied to a wide range of clinically relevant cell types including CD34+ hematopoietic stem cells (HSCs), primary T cells and NK cells.
These unique features make it a promising candidate to improve clinical transduction protocols for ex vivo gene therapies and CAR-T cell therapies.
LentiBOOST™ features include:
- Shows up to 90% improvements in lentiviral transduction efficiency.
- Positive impact on cell proliferation for CD34+ and T cells.
- Non-cytotoxic.
- Increased vector copy numbers per cell.
- Successful track record of integration into clinical trials (III and I/II) in the US and Europe.
- GMP batch grades available.
LentiBOOST™ benefits
LentiBOOST’s powerful features and advantages deliver a series of user benefits:
- Increased expression levels of therapeutic protein have favorable effects on success rates of clinical trials.
- Better-optimized and more stable transgenic cell manufacturing allows reduced cost of final product.
- LentiBOOST™ exhibits strong and durable therapeutic protein expression, in line with FDA/EMA criteria for ATMPs production.
- Proven lack of cell toxicity and prior clinical applications expedite IND filings.
- Availability of cGMP batch grades allow direct integration into clinical programs.
LentiBOOST variants
LentiBOOST™ is available in two versions:
- LentiBOOST™ Pharma grade: intended for use in preclinical research and process development only.
- LentiBOOST™ GMP grade: for use in clinical stage protocols, currently included in multiple Phase III and I/II clinical trials in Europe and North America.
Contacts
For quotes and detailed information on LentiBOOST™ technology, contact: [email protected].
For licensing options for Clinical/Commercial Use and Development, contact: [email protected].
Resources
Click on LentiBOOST Transduction Enhancer for detailed information.