By Pharmatrans SANAQ…
Pharmatrans Sanaq introduces high quality MCC SANAQ® material
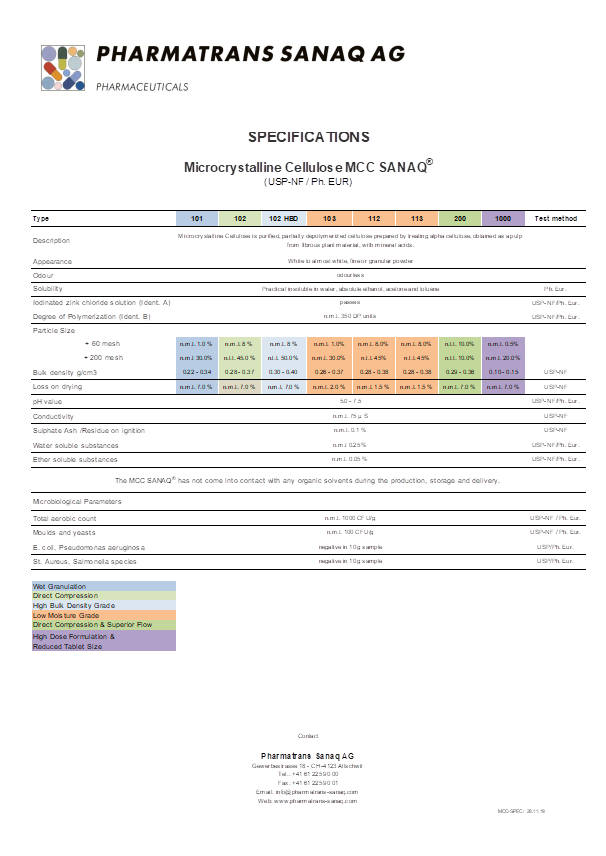
Pharmatrans Sanaq has introduced Microcrystalline Cellulose MCC SANAQ® as a highly purified and partially depolymerized a-cellulose with chemical and physical identities meeting the latest standards of Ph. Eur. and USP-NF European and American pharmacopeia standards.
The material features high standards in the selection of the raw material and the ingredients for production and strict observation of manufacturing GMP standards to ensure a fully compliant material.
MCC SANAQ® characteristics
MCC SANAQ® is a nonreactive substance with homogenous particle sizes. This makes it ideal as filter material in separation technology to guarantee high quality results.
The excellent compressibility and outstanding plasticity enable production tablets with high temper and ultimate strength, while retaining very short disintegration times.
Excellent flowability assists high production rates, while high microbial purity, color stability and excellent absorption properties of MCC SANAQ® contribute to success in all tableting processes.
MCC SANAQ® applications
MCC SANAQ® is mainly used as filling, binding and blasting material for tableting (direct tableting), as well as for dry and wet granulation and as filling material for hard gelatin capsules. It can also be used as filter material in separation technology. The material is also suitable for cosmetics, as stabilizer for emulsions, as absorption and turbidity material and as viscosity regulator. In the food industry, it can be used as a stabilizing system for both water and oil phase ingredients.
Resources
Click on MCC SANAQ specifications for detailed data.