By Dec Group
High potency API manufacturing solutions
API manufacturing requires powder handling in a Good Manufacturing Practice compliant (cGMP) and contained manner. Standard processes are similar to chemical manufacturing, including charging powders into reactors, emptying centrifuges or charging and discharging dryers. In addition, Dec’s suite of interconnectable systems provide solutions for Micronization/Milling, Dryer Discharge, Liquids Handling, Blending/Mixing, Dispensing, Sampling, Bulk Handling, Aseptic and Pack Off processes, all under a safe and controlled regime.
High Containment
The increasing potency of pharmaceutical ingredients, together with the ever-greater reach and rigor of regulation, dictate that potential hazards must be tackled at source, within the handling and manufacturing processes and by way of mechanical/process isolation to make operator personal protective equipment (PPE) redundant.
Dec Group meets these imperatives with a systems approach to powder and liquids handling and processing based on a proven track record in managing highly technical and challenging projects extending back more than 30 years.
The Dec systems approach emphasizes integration of new or existing equipment and provision of comprehensive safe, ergonomic and dedicated solutions achieving high containment down to the nanogram level.
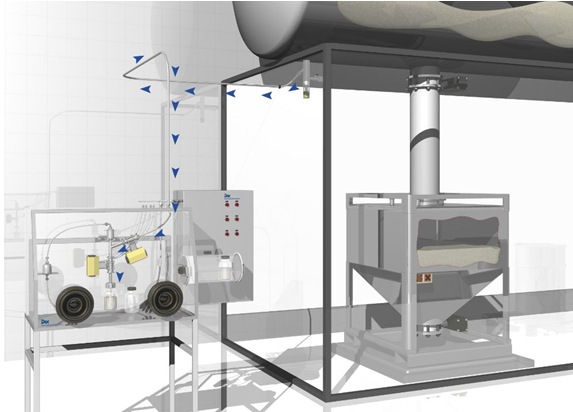
PTS technology for highly contained process linking
As an important component to high containment is PTS with its unique ‘push-pull’ approach to moving powders under total containment, using vacuum to draw powders into and through the system and pressure to push them into designated receptacles.
The PTS principle as applied to a series of designate process modules that can form an integrated high containment network covering the full range of process disciplines and challenges involved in high containment pharmaceutical API manufacturing.
API manufacturing solutions
Dec Group solutions for high containment API manufacturing include:
Liquids Handling: A complete system for charging liquids that are toxic, highly flammable or with strong odors from drums into reactors, based on the DCS® Liquid solution for filling and emptying drums, specifically developed for toxic and corrosive liquids, achieving containment levels of < 1 ppb. Safe and contained emptying and filling of drums is thus enabled without additional precautions such as full protective clothing or large laminar airflow booths. DCS® Liquid can be installed on load cells and be equipped with dosing valves allowing accurate weighing and dosing of the liquid.
Reactor Charging: Dec has developed a large range of products to guarantee full safety through high containment and oxygen elimination during the powder charging process that allow powders to be charged and dispensed directly out of various packaging such as bags, drums or big bags into the reactor, whereby the transfer rate can be controlled in case of exothermic reactions.
Aseptic Manufacturing: Dec systems enable to run a fully closed operation under strictly controlled conditions. At the forefront of both development and implementation of aseptic manufacturing solutions, Dec provides systems for various processes such as transferring, filling/dosing, dispensing, micronizing, cleaning including a whole range of process isolators and glove boxes.
Blending/Mixing: Dec’s patented PTS Batchmixer® technology can meet the most stringent requirements for mixing powders, based on a self-charging system that does not contain any moving or rotating parts. PTS Batchmixer® can be easily integrated in a production line and be the interface between the various operation steps. The process is therefore completely sealed and due to its high GMP and cleanability design, it can be used to handle highly potent and/or sterile products.
Micronizing/Milling: Dec can integrate various milling equipment into their process solutions including Dec’s crusher, UMS DecMill and Dec’s jet mill range. Dec’s core expertise is to mill powder in the low range, typically below 100 microns up to below 1 micron, either by using the Universal mill or the patented MC DecJet® product line. Dec’s flexible, multi-format spiral jet mills are designed to satisfy the full range of requirements and are available in open and contained arrangements to micronize difficult and hard to handle products.
Centrifuge Discharge: Dec has extensive expertise to transfer solvent wet powder directly out of a centrifuge (bottom or side discharge) and convey the powder directly to a dryer or back to reactor. When a direct transfer is not possible due to process timing or validation requirements, powder can be transferred into an intermediate silo before being transferred to the final destination. PTS® allows wet powders to be conveyed over more than 30 meters, while specially designed hoppers installed at the outlet of the centrifuge can handle very sticky or lumpy powders.
Dryer Discharge: Dec has developed a large range of dryer discharge solutions, accommodating various types, heights and containment levels. The filling station can be directly installed below the dryer to be discharged. In case of insufficient height under the dryer or when one single filling station needs to be shared between several dryers to be emptied Dec proposes their unique solution of combining both transfer and dosing in one single machine. The concept furthermore isolates the filling station from the dryer preventing any influences on the weighing system caused by pressure which may be present in the dryer.
Pack-Off Solutions: Dec’s pack-off solutions are similarly based on a unique unified transfer/dosing concept enabling final packing of APIs in a contained manner without the requirement of a clean room around the packing area.
Sampling: Dec provides various sampling devices from the vacuum type MPTS and DCS® Sampling for incoming materials in drums to a mechanical system to take samples inline directly from equipment such as dryers, mixers or from a pack-off line. The sampling procedure with MPTS is simplified, whether inline or out of process, from equipment with poor access, or where personnel are not admitted for safety reasons. Samples are extracted and transferred to final destination, whilst maintaining full containment, remaining homogeneous and representative as taken in dense flow, without using any mechanical device that could result in powder attrition.