By Gerteis Maschinen…
Gerteis® roller compaction for continuous manufacturing
Over more than three decades, Gerteis® of Switzerland has become the world’s leading name in in the roller compaction of pharmaceutical powders, particularly those demanding dry granulation for continuous production techniques.
Starting as a technology historically reserved for products sensitive to heat or moisture, products, dry granulation roller compaction has evolved to become a widely used powder processing technology with broad applications that now include continuous manufacturing of pharmaceuticals such as powders and tablets.
The dry granulate is either an intermediate product to be further processed into tablets, or it already represents the final bulk product to be packed in sachets, pouches, blister packs, plastic bottles, canisters, or used in the production of tablets and capsules.
Advantages of dry granulation
In contrast to wet granulation, dry granulation is a continuous production system. This results in various economic advantages, not least increased throughput with dry granulation systems reaching 400 kilograms per hour (kg/h).
The main purpose of dry granulation (DG) is to increase the bulk density of powders and increase the particle size to ensure a better flow of distributed material, in turn resulting in easier processability. In pharma, the goal is often to achieve a more uniform and therefore less friable tablet composition. DG powder blends can be more homogeneous, avoiding segregation and increasing content uniformity. Further, DG roller compaction also reduces dusts and fines, especially for bulk products, resulting in little or no material wastage and low dust exposure, which is particularly important when handling highly potent active ingredients (HPAIs).
DG roller compaction
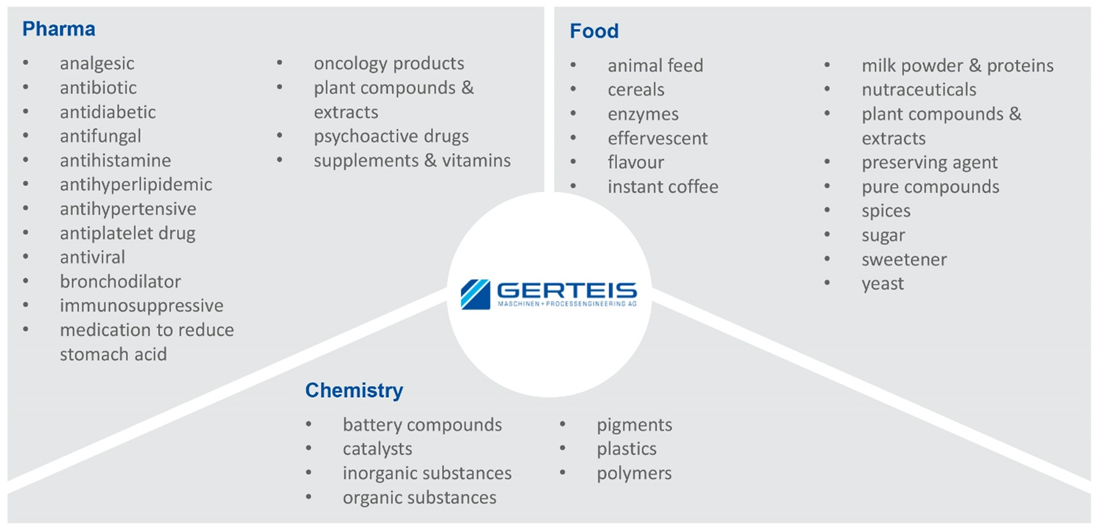
Variable gap roller compactors using sophisticated process controls that are specialty of Gerteis® are needed whenever granules of consistent and high product quality are required. While the lion’s share (~ 95%) of Gerteis® roller compactors go to pharmaceutical manufacturers, they are also demanded by other industries, notably food and chemicals for manufacturing a wide range of products (Figure 1).
The number of products being manufactured by Gerteis® Method Roller Compaction (RC) continues to increase steadily as the technology proves its advantages. It is continuous, has a relatively large throughput, requires little floor space, needs very little energy and benefits from the absence of granulation liquids and drying equipment.
Roller compaction can be applied to a wide range of pharma products. Many generic drugs are dry granulated, for example Metformin, Ibuprofen, antihypertensives, and antibiotics and proprietary products, including blockbusters such as Metformin and orphan drugs like Bosentan, are increasingly being developed for DG manufacturing.
Although roller compaction is a continuous manufacturing (CM) process suitable for both large and small batch sizes, almost all products are manufactured in batch format. The reason for this is that currently few products have been approved as a fully CM Pharma process. One example, Symdeko from Vertex was approved 2018 by the FDA having dry granulation integrated in a continuous manufacturing pharma line.
Roller compaction can also be used in the manufacture of effervescent formulations in one blend without separating the carbonate salts and the acid components. This reduces the effort of manufacturing two separate batches of wet granulate to a single manufacturing step.
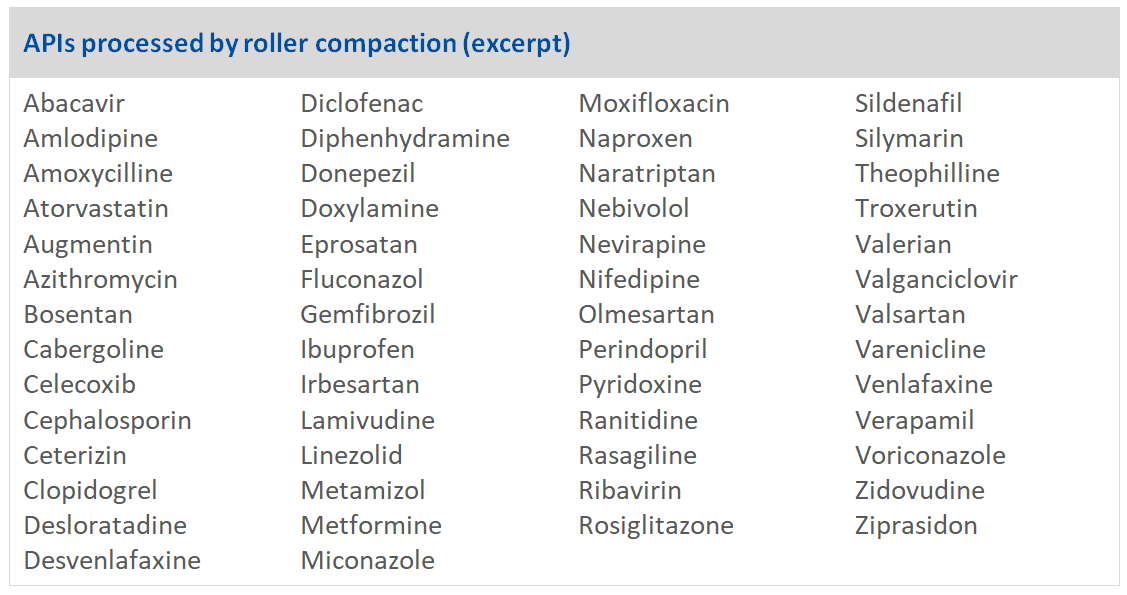
Even if a substance was originally developed as wet granulate, it is often possible to change the granulation method to dry granulation. A wide range of APIs are now processed by roller compaction (see Table 1), with successful changes from wet to dry granulation already performed for Metformin, Ibuprofen, Triamteren & Hydrochlorothiazide (HTCZ), with the latter demonstrating that candidates for roller compaction are not limited to single API products but can be API combinations.
Pharma roller compaction concepts
Granulation is a process in which powder particles are made to adhere to each other, resulting in larger, multi-particle entities; ‘granules’. In order to obtain the desired granules, the compaction process is followed by a milling step.
Whereas at wet granulation process liquids need to be added in dry granulation, the powder blend is compacted by applying a force onto the powder, which in general causes a considerable size enlargement.
The compacts thus obtained are called briquettes, flakes or ribbons.
An RC process does pose some challenges. Firstly, roller compaction formulations generally require higher amounts of excipients and therefore lower drug loading compared to wet granulation. The exact formulation also depends on the compactability of the API. Secondly, the powder blend compressibility may be compromised when forming granules that are subsequently compressed into tablets.
Ribbon density or solid fraction, defined as the mass of material per volume of ribbon, is commonly considered a critical quality attribute (CQA) of an RC continuous manufacturing process, requiring active measurement and control.
However studies have demonstrated that an RC design space for a broad range of throughputs can be predicted with a high accuracy. It may be used in the future as a soft sensor for RC processes operating in continuous manufacturing lines for in-line adjustment of throughput while maintaining the desired ribbon solid fraction. In order to calibrate the model for a new material in gap-controlled mode, it is necessary to perform the calibration routine to extract the compression profile and the relaxation factor.
Furthermore, it is only necessry to model a small fraction of the RC process in order to predict the ribbon density with high reliability, allowing for close prcess control.
Versatile Gerteis® RC technology
The versatility of renowned Gerteis® pharmaceutical roller compactors, including POLYGRAN®, MACRO-PACTOR® and MINI-PACTOR® support compaction of all kinds of powders, even the most difficult ones at all scales.
Dry granulation roller compaction has become a versatile, efficient, and time- and money saving method embraced by various industries that rely on consistent product quality and sophisticated process control. In the pharmaceutical industry, a large variety of formulations can be processed using roller compaction: batch or continuous, high-containment, proprietary or generic, blockbuster or orphan drug, high or low drug load, immediate or sustained release, API combinations, and many more.