By Evercyte GmbH
Evercyte Extracellular Vesicles (EVs)
Evercyte offers cells and services for projects involving extracellular vesicles (EVs) / exosomes that play an important role in cellular communication, due to their capacity to transport proteins, lipids as well as nucleic acids.
Specifically, extracellular vesicles secreted from human stem cells appear to be as effective as transplanted cells in various disease models.
What are extracellular vesicles?
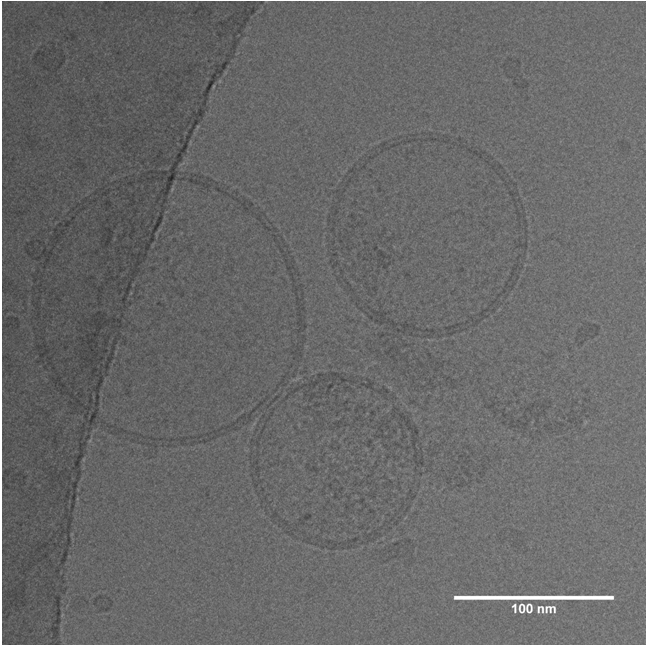
Extracellular vesicles are released from cells on fusion of a multivesicular body (MVB) intermediate endocytic compartment with the plasma membrane, thus liberating intraluminal vesicles (ILVs) to go extracellular.
They can contain a vast array of different proteins depending on their host cell, further modulated by cellular states such as stress, activation, or inhibition of specific signaling pathways. The most common canonical marker proteins are tetraspanins like CD9, CD63 and CD81, present on the vesicle surface.
Extracellular vesicles and MSCs in current research
Extracellular vesicles have become focuses in research for their role in intercellular transmission of macromolecules and in the spread of proteins, lipids, mRNA, miRNA and DNA as contributing factors in several diseases. Further, because they are composed of cell membranes and thus better tolerated by host, they seem to form useful vectors for drugs.
One of the most interesting group of extracellular vesicles is related to Mesenchymal stem cells (MSCs), the multipotent stromal cells that are found in various parts of the human body and which play important roles in maintaining tissue homeostasis and function. Several studies suggest that MSCs secrete extracellular vesicles that perform as mediators in the tumor niche and play several roles in tumorigenesis, angiogenesis, and metastasis. Other studies suggest tumor-suppressing effects of MSC-derived exosomes.
MSCs are a particular focus for research as they show a high degree of differentiation and exert immune modulatory properties through direct cell-cell contacts as well as secreting soluble factors, migrating towards sites of injury. Thus, MSCs carry an enormous therapeutic potential in developing novel clinical applications and EVs secreted from MSCs have been shown to exert immunomodulatory properties and anti-inflammatory or angiogenic properties Therefore, EVs could substitute for using cells in the therapy of various diseases, from graft versus host disease to autoimmune diseases.
EV isolation methods
EVs defy imaging by conventional methods, being released only sporadically and too small to be seen by fluorescence microscopy. It is possible to see EV release occurring in cell cultures using electron microscopy but the main method of isolation is to pool exosomes from cellular supernatant or animal fluids, using differential centrifugation or tangential flow filtration.
Exosome-related Evercyte services
Evercyte uses reactivation of telomerase to establish human highly differentiated cell lines from different organs and tissues for EV production. It can establish cell lines for production of clinical grade EVs as a one-stop-solution, encompassing tissue sourcing, ethical approval, primary cell culture, life-span extension, detailed characterization of cells and secreted EVs. Principal platforms include:
- Wharton´s Jelly-derived and bone-marrow derived MSCs established under xeno-free culture conditions, fully documented as innovative EV production cell line
- Production of recombinant EVs (recombinant cargo, recombinant surface molecules for targeting and / or for purification (e.g. using ‘Snorkel-Tag’)
- Characterization studies of EVs (miRNA profiling, lipidomics, proteomics, surface marker profiling, electron microscopy)
Evercyte offerings include:
- ASC/TERT1 – telomerized human adipose-derived mesenchymal stem cell line with a differentiation potential similar to the corresponding normal cells.
- WJ-MSC/TERT273 – telomerized human Wharton´s Jelly derived MSC line represents a new production system for extracellular vesicles.
- BM-MSC/TERT277 – telomerized human bone-marrow derived mesenchymal stem cell line represents a new production system for extracellular vesicles.
Research applications include gaining new insights into the role of EVs in inflammation and wound healing, testing their potential of EVs in influencing the immune response, analysis of EV cargo and targeting and generation of recombinant EVs for purposes such as drug targeting
Exosomes and IPSCs
EVs are also secreted from induced pluripotent stem cells (IPSCs), exerting protective effects by transferring endogenous molecules to salvage injured neighboring cells by regulating apoptosis, inflammation, fibrosis and angiogenesis.
Exosomes from iPSC-derivatives are indicated to have therapeutic potential in the treatment of Cardiovascular disease (CVD), for example, with the emergence of iPSC)s and iPSC-derived cardiomyocytes (iCMs) presenting apparent opportunities to replenish the heart’s functional cells.
Evercyte has developed protocols for the isolation and expansion of cells isolated from fresh urine as well as frozen urine sediments. Both cell preparations show a 100% reprogramming efficiency and generated induced pluripotent stem cell lines express typical markers and functions of pluripotent stem cells.
Research applications include establishment of cell models across a broad panel of human diseases and development of differentiation protocols.