By Evercyte GmbH
Evercyte supports study into extended yeast display libraries for CD81 LEL mutants for EV surface customization
Vienna, Austria: – Innovative biotech manufacturer Evercyte has helped support and contribute an important study that establishes yeast display libraries of CD81 large extracellular loop (LEL) mutants selected towards specific antigen binding and resulting mutants could be used for custom functionalization of the EV surface.
Extensive collaboration
The study ‘Construction of Yeast Display Libraries for Selection of Antigen-Binding Variants of Large Extracellular Loop of CD81, a Major Surface Marker Protein of Extracellular Vesicles’1 was lead-authored by Dr. Stefan Vogt, a research fellow of the Austrian Centre of Industrial Biotechnology (ACIB) in Graz and the Institute of Molecular Biotechnology at BOKU University of Natural Resources and Life Sciences, Vienna, with a research team drawn from four other leading Austrian research institutions and including Evercyte’s co-founder and Scientific Advisor, Assoc. Prof. Dr. Johannes Grillari. The study now form part of the new book series Methods in Molecular Biology.
Evercyte co-sponsored the study in partnership with Vienna’s BOKU University of Natural Resources and Life Sciences, whose Institute of Molecular Biotechnology and Christian Doppler Laboratory for Innovative Immunotherapeutics were both closely involved in the research. Additional funding support was received from Austria’s FFG-COMET Funding Program.
Antigen recognition technology
The study was based on Evercyte’s patented technology of antigen recognition via antigen binding marker proteins of EVs and sought to establish a new model for yeast display methodology, which has become a leading tool for discovery, humanization, stability improvement, and affinity maturation of antibodies and antibody fragments, as well as for the development of diverse non-antibody protein scaffolds towards the ability of antigen recognition.
It also builds on an earlier study, also led by Dr. Vogt that established an engineered CD81-based combinatorial library for selecting recombinant binders to cell surface proteins, with laminin binding CD81 enhancing cellular uptake of EVs2.
A new yeast display model
Yeast display is particularly well suited for multiparametric analysis of properties of derivatized proteins, allowing the evolution of most diverse protein structures into antigen binding entities with favorable expression, stability, and folding properties.
The research establishes methodological basics for a novel yeast display-based approach for the functionalization of the large extracellular loop of CD81 into a de novo antigen binding unit.
Importance of CD81
The surface marker protein CD81 is intrinsically overrepresented on the surface of EVs as naturally occurring nanoparticle units that act as cell-to-cell messengers by delivering their intracellular cargo from the source cell into a recipient cell. While this amazing property of EVs makes them of primary biotechnological interest, methods for their targeted delivery are still in their infancy.
The unique EV membrane composition has for a long time influenced the choice of methods of EV identification and characterization. Current isolation methods rely on size-exclusion chromatography, ultrafiltration liquid chromatography, or affinity chromatography-based methods to ensure biophysical integrity of the final product.
With rapid expansion of knowledge over the past decade about EV bio-composition, the overrepresentation of certain membrane proteins has remained a characteristic feature of these particles. These include TSG101, Syntenin, and Alix, involved in ESCRT complex, functional in multivesicular body biogenesis, and most notably members of tetraspanin family: CD81, CD9, CD63 and CD151, contributing to the formation of microdomains, essential for exosome functional properties.
The most characteristic stability feature conserved among the tetraspanin proteins is the 4-cystein motif resulting in two invariant disulfide bridges, which connect the C-terminal region of the B-helix with the first turn of the E-helix and the BC-segment to the CD-loop.
Based on its significant overrepresentation in EVs, the team chose CD81 to be modified into an antigen-recognition scaffold.
Modified peptide grafting
Peptide grafting was the first method applied to experimentally identify the regions permissive for mutagenesis.
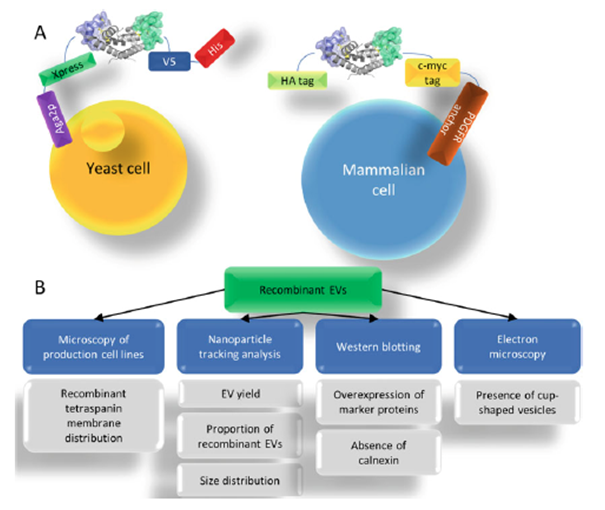
The proteins were expressed as soluble entities in mammalian expression systems and examined in parallel for the level of display on yeast cells. This method leaves open the future prospect to create yeast display libraries of randomized CD81 LEL that can be applied for discovery of antigen-specific binders.
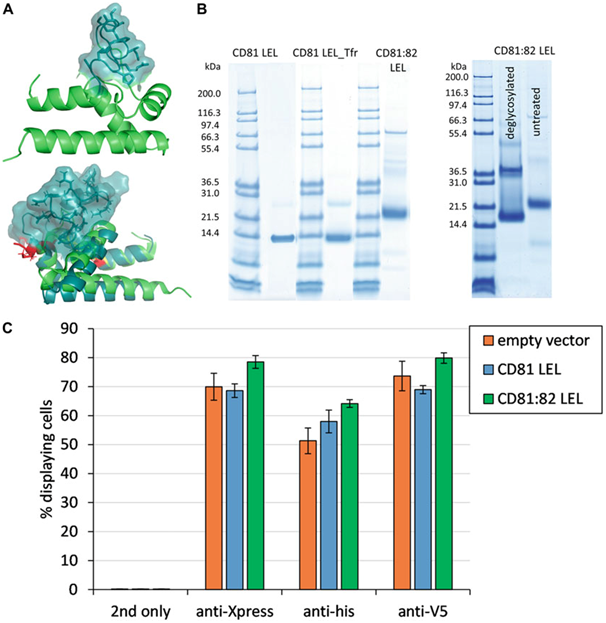
For simple peptide grafting, different locations on CD81 LEL were chosen and discovered to be differently amenable for this kind of modification. While the same peptide inserted into the loop connecting A and B helices caused reduced expression of the mutant protein, which displayed an aberrant profile in size exclusion chromatography (SEC) in native conditions, the mutant harboring the insertion into helix D expressed well and its elution profile was indistinguishable from the wild-type CD81 after preparative SEC. As the conformation-dependent antibodies used for corroboration of proper folding of CD81 recognize the epitope modified in this graft, it was not possible to judge on the folding properties of the mutant. Taken together, the simple peptide graft into D-helix was accommodated successfully enough to encourage an attempt of more extensive mutagenesis: the sequence pertaining to the variable region of class 2 tetraspanin protein CD82 was inserted at the positions encompassing the analogous structural unit in CD81 into the conserved sequences of CD81 protein. The resulting molecule could be purified from mammalian cell culture supernatant and displayed the expected molecular weight as judged by SDS-PAGE analysis.
Additionally, the N-linked glycosylation motifs characteristic for CD82 were utilized as peptide: N-glycosidase F treatment caused a decrease in molecular weight for about 5 kDa. The analysis of the hybrid molecule on the yeast surface infers the same level of display as established for the wild-type CD81.
This series of experiments led to important insights in the possible extent of CD81 engineering, confirming the possibility of inserting relatively long structured stretches of amino acids that offer a large surface amenable to further randomization, implying the possibility of a larger antigen interaction surface.
Conversion to EV Surface Molecules
Prior to conversion of candidate antigen binding LEL mutants into a full-length CD81 protein displayed on EV surface, which involves time-consuming development of stable transfected cell lines, false hits can be excluded by a simple screening step involving transient transfection of mammalian cells. Mammalian pDisplay vector enables N-terminal anchoring of the protein of interest as opposed to C-terminal fusion as present in yeast display vectors and features other expression tags, thereby minimizing the likelihood of binding artifacts without loss of sensitivity as the copy number of the displayed CD81 LEL molecules amounts to about 20,000 in both systems. The procedure from cloning to the final read-out requires 4–5 days, making this a viable tool for validation.
Following cloning into the full-length CD81 format and stable expression in the recombinant cell lines, the resulting EV preparations can be tested for certain properties. Transformed cell lines should exhibit the expression level and membrane distribution of CD81 similar to cell lines expressing the wild-type molecule.
Further, the overall yields, the proportion of recombinant particles and their size distribution should be comparable. Protein profiling should demonstrate overrepresentation of TSG101, syntenin, Alix, and absence of calnexin as a marker of apoptotic bodies.
Future Development of CD81 LEL for antigen recognition
In the first attempts of derivatization of CD81 for antigen binding, the confirmation of positive hits relied on internalization of identified binders into antigen-positive cell lines. However, for the establishment of a successful discovery platform, a definition of the required target profile should include the information on target affinity optimal for a particular biological situation, taking into account the biodistribution and bioavailability of the EV displaying the selected binding entity.
The family of tetraspanins includes several members with different degrees of sequence homology, and therefore it is likely that other molecules sharing the basic fold with CD81 will sustain similar modifications towards de novo antigen binding. CD9, another class 1 tetraspanin, is also strongly overrepresented on EV surface and its overexpression is reported to enhance the number of secreted EV particles. CD9 LEL is well displayed on the surface of yeast cells, is also amenable to thermal stabilization, can be well expressed, and hence renders itself well for a design of a de novo binding surface. The engineering of another antigen specificity into such molecule can upon simultaneous expression with antigen binding CD81 aid construction of a bi-specifically targeting EV.
Bispecific targeting offers advantages that include superior selectivity and novel biology phenomena such as potentiated target receptor internalization and degradation that can result from bi-paratopic binding. Bispecific and multi-specific EVs can thus open the way to innovative EV-mediated therapies, offering superior safety and efficacy compared to entities addressing a single target.
Mammalian Expression of selected library clones
The team applied thermally stabilized CD81 LEL mutants to examine the correlation between the apparent thermal stability of yeast displayed CD81 LEL molecules and the actual Tms determined by DSC. Although researchers established the same ranking of Tms determined by both methods, the properties of soluble and yeast displayed proteins did not match exactly: for example, heating of the mutant C2C9 in soluble form is reversible while the yeast displayed molecule appeared denatured when exposed to high temperatures.
Further data on thermostability properties of antigen binding variants as well as entire libraries will offer more insight in the destabilization effects of applied mutagenesis methods, leading to improved library designs and paving the way to superior stable mutants isolated by directed evolution.
References
- Vogt, S., Stadlmayr, G., Stadlbauer, K., Stracke, F., Bobbili, M.R., Grillari, J., Rüker, F. and Wozniak-Knopp, G. (2022). Construction of Yeast Display Libraries for Selection of Antigen-Binding Variants of Large Extracellular Loop of CD81, a Major Surface Marker Protein of Extracellular Vesicles. Methods in Molecular Biology, pp.561–592. doi:10.1007/978-1-0716-2285-8_24.
- Vogt, S., Bobbili, M.R., Stadlmayr, G., Stadlbauer, K., Kjems, J., Rüker, F., Grillari, J. and Wozniak‐Knopp, G. (2021). An engineered CD81‐based combinatorial library for selecting recombinant binders to cell surface proteins: Laminin binding CD81 enhances cellular uptake of extracellular vesicles. Journal of Extracellular Vesicles, 10(11). doi:10.1002/jev2.12139.
About Evercyte GmbH
Evercyte is the leading provider of immortalized human primary-like cells (‘telomerized cells’) as well as novel production cell lines for extracellular vesicles and is the partner of choice for innovative cell-based assays in the field of pharma, biotech, or cosmetic industries.
The core technology used for the establishment of these highly relevant human cell lines relies on the reactivation of the human telomerase enzyme. Besides customer-tailored cell line development as a one-stop shop, Evercyte is steadily expanding its catalog of readily available cell lines for the production of extracellular vesicles from mesenchymal stromal cells using different tissues including adipose tissue, bone marrow, placenta, amnion or dental pulp.
By providing native extracellular vesicles from mesenchymal stem cells and targeting extracellular vesicles Evercyte will make a significant contribution to the development of new therapeutic approaches. Additionally, Evercyte will promote its platform technology for the generation of recombinant EVs e.g. tissue targeting and targeted drug delivery.
Founded in 2011, the Vienna-based company successfully pioneered the development of tools and know-how necessary for the establishment of standardized high-throughput in-vitro bioassays as well as relevant cell models and cell factories.
Learn more at: http://www.evercyte.com.
Resources
Click on Construction of Yeast Display Libraries for Selection of Antigen-Binding Variants of Large Extracellular Loop of CD81, a Major Surface Marker Protein of Extracellular Vesicles to access the full study.
Click on Evercyte engineering of CD81 on extracellular vesicles to target specific cell surface proteins to learn more.
Click on Evercyte recombinant and engineered exosome services for further information.