By Evercyte GmbH
Evercyte Extracellular Vesicle (EV) Potency Testing
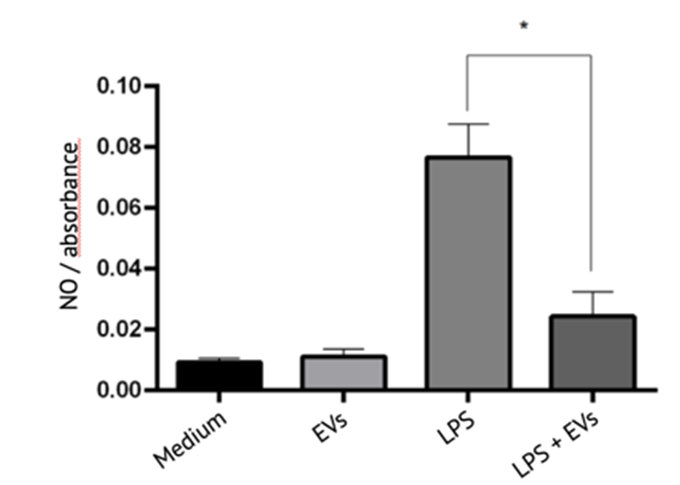
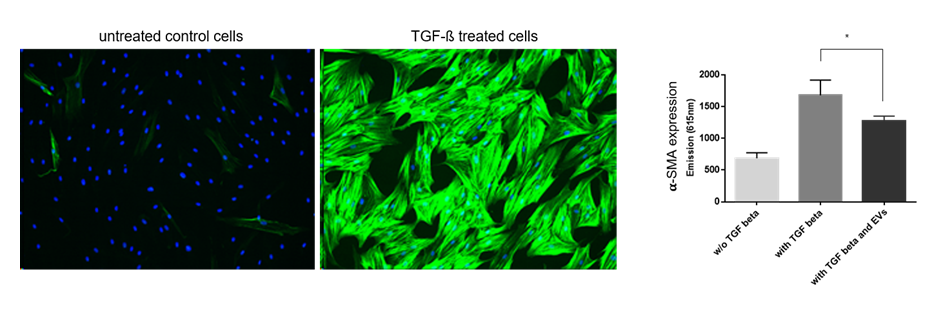
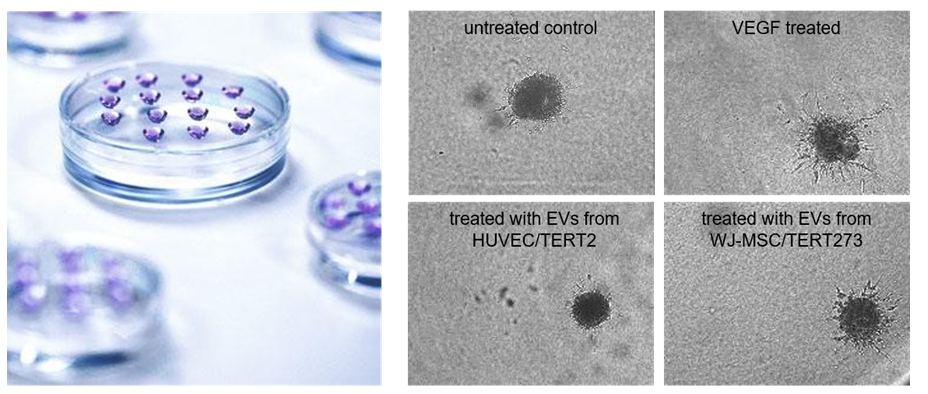
Evercyte can offer services to test the potency of extracellular vesicles (EVs) and exosomes across a wide range of applications, including anti-inflammatory, anti-fibrotic, wound healing and neo-angiogenic therapies.
These services are distinctive in using human relevant and standardizable cells from different organs and tissues as the basis for testing EV biological activity.
Meeting therapeutic needs
Extracellular vesicles, including exosomes, are secreted by human mesenchymal cells and exert biological activity.
As EVs and exosomes gain in status as new therapeutic options for unmet medical needs, testing their potency in specific therapeutic applications has become increasingly important.
Evercyte has established protocols for isolating specific EVs, characterizing them and in assisting its customers to developing EV therapeutics. It can also offer EVs from different mesenchymal stem cell lines for standards testing or in vitro assays.
Evercyte also offers EV production cell lines that will allow the production of clinical grade EVs.
EV testing service features
Evercyte’s EV testing services cover a wide range of options and needs. These include:
- Enrichment of EVs from cell culture supernatant
- Characterization of EVs for presence of EV marker proteins, typical morphology, miRNA cargo, etc
- Characterization of in vitro biological activity
Service benefits
Evercyte scientists have world-leading experience in the fields of EV development and characterizations, having amassed more than 25 years of work and experience with cell line development and the establishment of relevant and standardizable in vitro bioassays as well as EV production and potency assays. The company’s scientists have published more than 130 peer reviewed articles in these fields.
This means excellent data quality is assured as customer derived samples, such as EVs, cell culture supernatants or EV producing cells, are handled by very experienced cell biologists with in-depth expertise in handling and culture of cells from different origins using 2D as well as 3D cell culture systems, down-stream processing of cell culture supernatants such as EV enrichment or purification and in biochemical / biological characterization of EVs as well as EV production cell lines.
Evercyte also has access to the whole spectrum of techniques and reagents required for EV enrichment and characterization as well as specialized EV production cell lines and systems for lab scale EV production.
They can carry out relevant and standardizable in vitro bioassays using Evercyte telomerized cells that are closely matched to application.
Crucially, Evercyte can offer fast-track testing, using pre-established and validated techniques are established and assays performed without delay.
In all these ways, Evercyte EV potency testing meets the real-world needs of the biotech and chemical industries, pharmaceutical companies, SMEs, clinical research groups, start-up companies, research organisations, and academia.
Resources
Click on Evercyte Services testing the potency of EVs for further information.