By Evercyte GmbH
Evercyte EVs from human bone marrow-derived MSC (EV-BM-MSC/TERT292)
Evercyte can now offer extracellular vesicles (EVs) from human bone marrow-derived mesenchymal stromal cells (MSCs) as the latest addition to its range of ready-to-use off-the-shelf EVs derived from a variety of different human MSCs.
Extracellular vesicles derived from human bone marrow MSCs (EV-BM-MSC/TERT292) meet the life sciences industry’s need for ever-more tailored MSCs as evidence accumulates of the therapeutic effects of MSC are in part mediated by their secreted EVs.
Enhanced life and differentiation
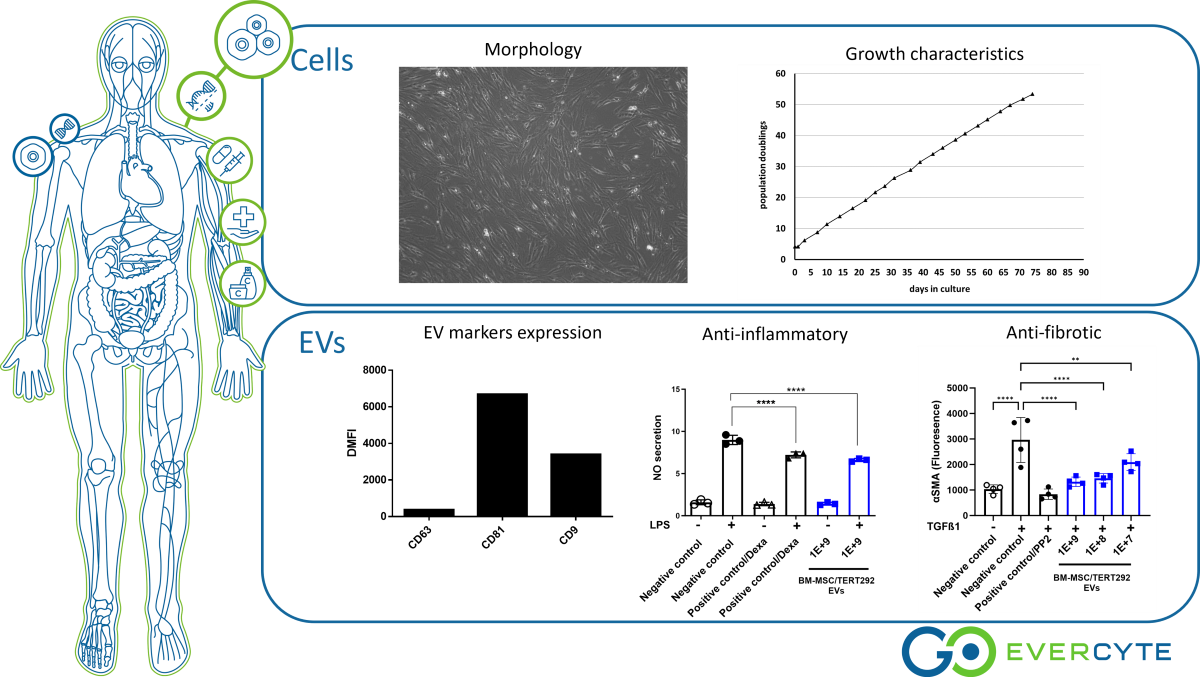
By overexpression of the catalytic subunit of human telomerase (hTERT), Evercyte can extend the cellular life span of various cell types (‘telomerized cells’) while maintaining their primary cell-like characteristics. Evercyte’s latest telomerized BM-MSC/TERT292 cells show typical markers and functions of primary MSCs with the added advantages of longer life spans and retention of tri-lineage differentiation, making them superior platforms for EV production and clinical applications.
Similarly, EVs derived from BM-MSC/TERT292 maintain typical EV-marker expression profiles and morphological characteristics, show anti-inflammatory and anti-fibrotic activities.
Clinical advantages
Evercyte also offers ready-to-use EVs from different mesenchymal stromal cell sources e.g. Wharton’s Jelly, adipose tissue and placenta. It has established protocols for 2D and 3D EV production, isolation, characterization and in vitro potency assays, and can assist its customers in developing EV therapeutics based on their needs.
Evercyte production host cell lines are optimized to support production of clinical GMP-grade EVs, with features that include:
- establishment following ethical standards (approved by IRB) with prior given written informed consent for commercial use of the isolated cells
- full documentation of all manipulation steps and certified xeno-free culture conditions
- quality tested for sterility, absence of specific human-pathogenic viruses, STR-profile, longevity, etc.
- characterized for growth, phenotype and presence of cell type-specific markers and functions.
Excellent data quality is assured as Evercyte’s scientists have world-leading experience in the fields of EV development and characterizations, having amassed more than 25 years of work and experience with cell line development and the establishment of relevant and standardizable in vitro bioassays as well as EV production and potency assays.
Resources
Click on Product page for further information.
Click on Evercyte EV production cell lines for further information.
Click on Evercyte ready-to-used EV advantages for further information.
Click on Evercyte EV services to learn more.