By Erdmann Solutions…
Erdmann’s human centered design applied to Trajan HemaPEN blood microsampling device
Erdmann Design AG successfully applied its HCD (Human Centered Design) methodology to the design and introduction of a revolutionary diagnostic medical device. The HemaPEN from Australian-based Trajan Scientific & Medical.
The process focused on user-centric testing and analysis to maximise HemaPEN’s market appeal as an easy to use solution.
HemaPEN concept
The Australian company Trajan Scientific and Medical develops technologies for scientific analysis systems for biological and food-related measurements. Trajan had developed the HemaPEN concept as a simple and reliable tool for Medical Practice Assistants (MPAs) to use when collecting microsamples of capillary blood for testing.
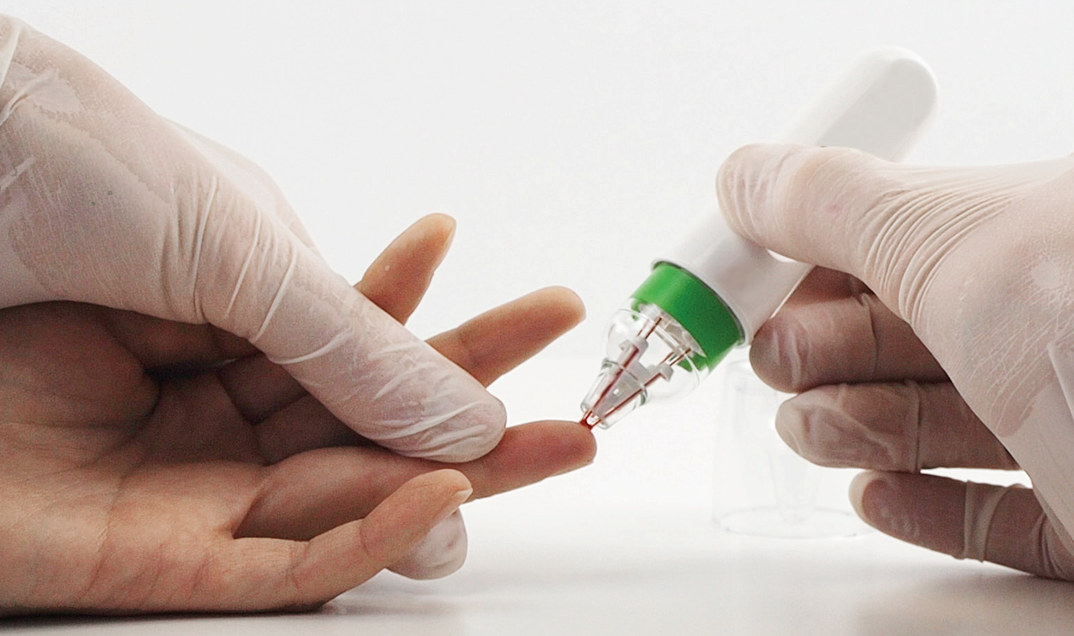
The HemaPEN consists of four capillary tubes and a sealable storage unit that guarantees safe transport of collected blood samples to the laboratory.
The second phase of development aimed at bringing the same solution to the market for private individuals, patients and their carers to use in home testing.
Starting from usability
Erdmann Design identified opening up homecare market with a device suitable for non-professionals as the crucial goal to guide the project. This means a rigorous focus on ease of use as the foundation of the design process, starting with a usability evaluation and a group-driven learning process in handling the HemaPEN and how to simplify it.
Results from the studies were tested with specialists from the medical field and prepared for the Trajan product development team. Documents created and consequent design improvements also benefited the company is achieving more rapid FDA and CE market approval.
Human-centered Testing
The Product context, including user aspects and application, was thoroughly explored and positioned with the firm in workshops.
The stage was set with in-hospital testing, carefully planned in mixed user-groups, segmented by age, education, experience, and roles of the participants, with camera-equipped individual observation stations placed at key process points. Resulting video recordings were shared with all participants, along with a set of questionnaires to establish the views and priorities of different user groups, including Wet Lab, Dry Lab, MPAs, patients and quality assurance (QA) professionals. Questionnaire results provided a further feedback loop to drive further product development and marketing.
In support of the process, Erdmann produced a product function film with didactic animations, a QuickGuide with easy to understand instructions for action.
HemaPEN sub projects have included Product Development Industrialization, usability testing, FDA and CE approval documentation, didactic concept development, cinematic animation and print media.
Resources
Click on Erdmann Design Usability Testing for further information.
Click on Trajan Scientific & Medical HemaPEN for product details.
Click on Experience Mapping for Human Centered Design to download PDF.