By Erdmann Solutions…
Erdmann Design helps develop hemaPEN® coronavirus testing solution
Brugg, Switzerland:- Leading edge design and engineering agency Erdmann Design has played a key role in developing the innovative hemaPEN® SARS-Cov-2 testing solution from Trajan that has now been submitted for regulatory testing and approval.
Erdman has supported the project with prototype development, usability testing as well as developing value propositions and risk analyses with clinical testing to support regulatory approval.
COVID-19 response
The COVID-19 pandemic has highlighted urgent need for new analytical tools and methods, to better understand SARS-CoV-2 related viruses and develop suitable vaccines and disease therapies.
Trajan Scientific and Medical has partnered with Erdmann Design to develop and validate an innovative, market-leading SARS-CoV-2 serology assay, available for high-throughput clinical research applications.
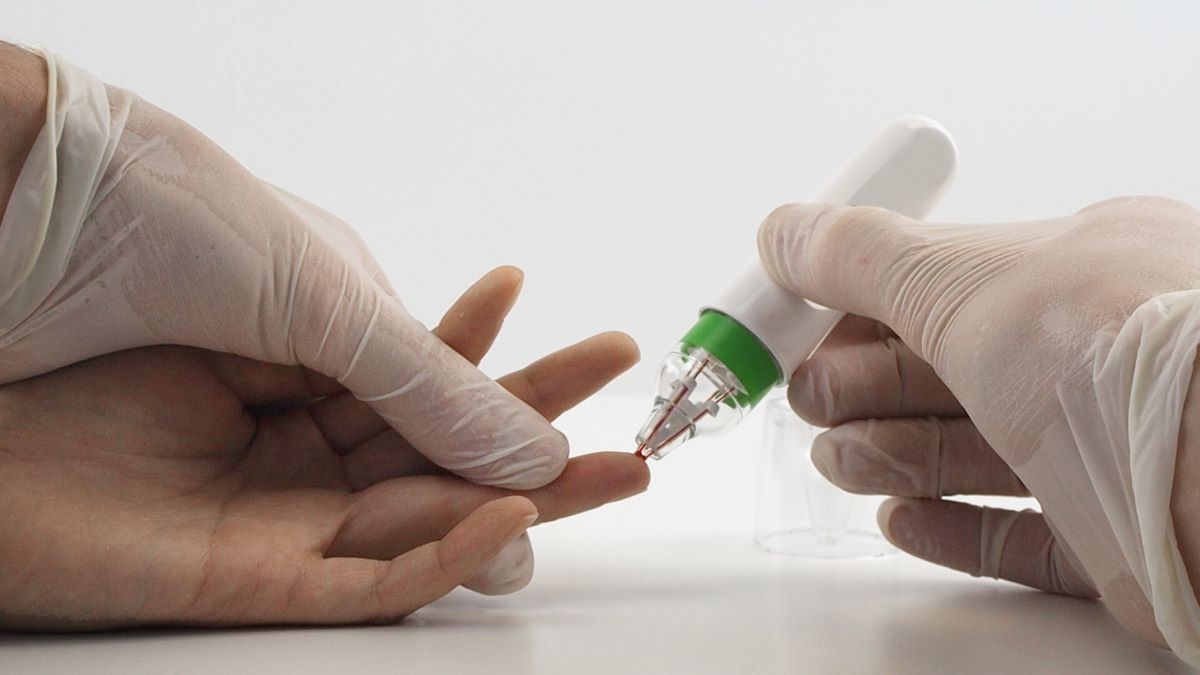
Microsampling technology
The resulting hemaPEN® tool uses Trajan’s advanced precision microsampling technology in conjunction with Synexa’s innovative, market-leading SARS-CoV-2 serology assay that is capable of detecting IgG, IgM, IgA and nAb antibody response, as well as their semi-quantification without the need for titration.
Together, these technologies deliver a highly competitive and flexible assay, suitable across a wide range of applications that can expedite therapy and vaccine development as well as large-scale screening, sero-surveillance and post-market surveillance initiatives.
Supporting vaccine roll-out
The tool incorporates innovative, easy-to-use design and microsampling technology and is aimed to support large scale screening and sero-surveillance studies post-vaccine roll out that can be conducted safely and efficiently, maintaining patient-centricity, comfort and convenience while observing social distancing protocols.
“We are proud to have supported Trajan is delivering this valuable project,” commented Erdmann Design founder and CEO, Raimund Erdmann.
“Through this collaboration, we have helped them develop a powerful testing platform supporting vaccine programs with more convenience and comfort in packaging and handling for the patient, while harvesting high quality safety and efficacy data,” commented Mr. Erdmann.
About Erdmann Design
Erdmann Design AG (Erdmann) offers award-winning human-centered Design and Engineering services that are particularly applicable to Life Sciences and Medical Precision Technology applications including pharmaceutical devices, patient monitoring systems, delivery robots, production systems and environmentally friendly packaging solutions.
The Erdmann Design Approach are built around Innovation, Human Centric Design underpinned by usability engineering and testing and Branding Expertise, bringing tightly knit and dynamic design teams to focus on innovation and real customer needs, with particular expertise in high-risk in technologies in Medical and Pharmaceutical Procedures.
Erdmann Design was founded in Brugg, in the Swiss canton of Aargau, close to Zurich, in 1978 by industrial designer Raimund Erdmann, who still leads the company. Since then, it has won more than 65 outstanding awards for its work with companies worldwide.
Further information at: https://erdmann.ch/
Resources
Click on Trajan/Synexa hemaPEN® SARS-CoV-2 testing for further details and product brochure.
Click on Erdmann Design Experience Mapping for more on Human Centered Design.
Click on Swiss Testing for Down Under for further details about the usability testing.